Oversight Gaps and Conflicts
Executive Summary
Every day in the U.S., 33 people die because they don’t have access to an organ transplant, and yet the nation’s organ donation system is leaving behind tens of thousands of viable organs.
Organ donation reform is more urgent now than ever before, given evidence that COVID-19 increases the risk of organ failure. Failures within the U.S. organ donation and transplantation system – which disproportionately harm patients of color – are left unaddressed by oversight bodies. This report examines the current oversight structure and provides recommendations to save more lives through organ donation.
“Failures within the U.S. organ donation and transplantation system – which disproportionately harm patients of color – are left unaddressed by oversight bodies.”
None of the 57 Organ Procurement Organizations (OPOs) operating in the U.S. have ever been successfully decertified by the federal government, even OPOs with:
- a 1 in 4 recovery rate of potential donors;
- leaders who went to federal prison for a tissue procurement kickback scheme;
- a failure to report a fungus that nearly killed a recipient; and, separately, a supervisor who allegedly physically assaulted a nurse and doctor; and
- countless other issues that have led to unrecovered, mishandled, lost, or damaged organs; patient deaths; and inappropriate use of taxpayer money, sparking Congressional investigations into fraud, waste and abuse.
We found oversight gaps or conflicts in 6 key areas:
- OPO failure to recover enough organs: OPOs can easily hide or manipulate their outcome measures in the current system, leading to thousands of organs going unrecovered and/or untransplated, and patients needlessly dying on the waitlist. This compromises not only oversight and regulation, but also academic research, which would ideally function as a means to inform ongoing policy considerations.
- Complaints process: OPOs are expected to self-report patient safety issues to the federal contractor tasked with optimizing their performance, the United Network for Organ Sharing (UNOS). But, even in UNOS’ determination, the voluntary nature makes these reports “subject to underreporting.” We found that few complaints are ever lodged against OPOs due to a lack of confidence that change will occur, fear of retribution if someone makes their concerns known, and unawareness of the reporting process.
- Conflicts of interest: Committees and boards within UNOS, as well as reviewers at academic journals, are often filled with OPO leaders, board members, and/or other member representatives. This leads to situations such as leaders from underperforming OPOs deciding their own performance review process or suppressing unfavorable research.
- Communication across a diffuse government structure: The groups within the Department of Health and Human Services (HHS) that ultimately oversee the organ donation system are reported to have a lack of communication and collaboration.
- Financial incentives: The large gap in oversight of financial incentives and resource allocation has led to issues such as OPOs spending a disproportionate amount of resources on lucrative tissue procurement, and OPOs spending taxpayer money on things like football tickets and lobbying expenses, while under-resourcing key programming more likely to lead to life-saving organ recoveries, such as hiring and supporting frontline staff.
- Technology and security: UNOS technology is outdated, inadequate, and insecure, yet government oversight arms are not auditing it, managing it in any meaningful capacity, or otherwise holding it to an appropriate standard.
In order to realize the potential impact of the new Final Rule for OPO performance evaluation that HHS published in November 2020 — which is projected to result in more than 7,000 more lives saved every year, while saving $1 billion annually to Medicare — we believe it is necessary for HHS to make parallel reforms to oversight and governance. We believe a new, empowered Office of Organ Policy (OOP) could be the ideal structure to oversee implementation of HHS’ new accountability measures and other crucial reforms. We also recommend decreasing conflicts of interest, improving the existing survey and complaint process, and increasing transparency and data visibility to monitor stakeholder compliance with federal regulations as well as foster more competition and innovation. In parallel, Congress should play an essential role in OPO and UNOS oversight to support and inform actions taken by the OOP.
Introduction
Each year, as many as 28,000 viable organs fail to reach patients who critically need them.1 Because of the failure to recover and transplant those organs – and the continued pace of demand outstripping supply – nearly 33 people die every day because they don’t have access to an organ transplant.2
Organ procurement organizations (OPOs) are responsible for working with hospitals to recover as many organs from deceased patients as possible and to find a suitable recipient while the organ is still viable. There are 573 such OPOs spread throughout the U.S., and their performance differs dramatically.4
According to one analysis, some of the lowest performing OPOs – such as the federal contractors in Kentucky and South Carolina – recover organs from only 1 in 4 potential donors.5 This underperformance translates directly to patients dying unnecessarily on the organ waiting list, with the burdens of OPO failures falling disproportionately on people of color.6
However, none of these OPOs has ever been successfully decertified by the federal government. OneLegacy, the country’s largest OPO, was deemed, “one of the worst performing OPOs in the country” by Rep. Katie Porter.7 8 The Los Angeles-based center recovered organs from only 31% of potential donors, according to an analysis from the University of Pennsylvania on OPO performance from 2012 to 2014.9 Despite the OPO’s underperformance, OneLegacy’s CEO Tom Mone is paid more than $904,000 per year10 and its Board Chair is paid $100,000 annually with taxpayer money.11 The group also misspent more than half a million taxpayer dollars on “unallowable or poorly documented items” – including football tickets, parade expenses, and lobbying costs – according to a government audit.12 13 14 Subsequently, a Los Angeles Times investigation found OneLegacy to have interfered with death investigations through improper tissue harvesting and then to have coordinated with coroners’ offices to cover it up.15 16 Even more recently, in December 2020, OneLegacy was the subject of a Congressional oversight inquiry from the House Committee on Oversight and Reform seeking information regarding potential conflicts of interest, executive and board compensation, anti-patient lobbying, and the OPO’s use of Paycheck Protection Program loans.17
OneLegacy is far from the only OPO tainted by scandal. In Alabama, OPO leaders were sent to federal prison for participating in an illegal kickback scheme with a local funeral home.18 The former director and associate director of Alabama Organ Center (which has since re-branded to “Legacy of Hope”) pocketed nearly $500,000 in the plot involving lucrative tissue harvesting.19
In South Florida, a doctor and nurse alleged being physically assaulted by a manager of an OPO for speaking up about personnel shortages and inadequate training.20 The two women were let go; one was fired, and the other’s contract was not renewed after 17 years of working there. Meanwhile, the supervisor kept his position of leadership.21 Four years later, the same OPO nearly killed a woman for failing to identify a fungus on a uterus before sending it out for transplantation — and did so during a period in which the OPO was already deemed a “member not in good standing” due to a “serious lapse in patient safety or quality of care.”22
In South Carolina, the OPO We are Sharing Hope SC shipped lungs and a liver with an incorrectly identified blood type – killing the lung recipient and injuring the liver recipient.23 The Post and Courier, a South Carolina newspaper, noted that it was only after the news outlet started asking questions about We are Sharing Hope SC that oversight agencies said they would review the case. When investigators finally looked into the South Carolina OPO after the incident garnered news attention, they found problems with We are Sharing Hope SC’s blood-typing policies.24
Despite systemic issues throughout the organ donation system, there has not been extensive scrutiny of OPOs by oversight agencies. Additionally, there’s a lack of competition to drive performance, since OPOs operate as monopolies in their respective regions. The Organ Procurement and Transplantation Network (OPTN) was created to oversee OPOs and coordinate organ allocation throughout the country. However, only one contractor has ever held this position, the United Network for Organ Sharing (UNOS), and they are also rarely held accountable by federal oversight agencies. Historically, all OPO data shown to public and regulatory bodies – including reports of patient safety issues and failures to recover viable organs – have been self-reported to UNOS, allowing OPOs to mask errors and poor performances.
A new rule proposed by CMS in 2019 and finalized in November, 2020, offers new objective measures to better assess OPO performance and provides a pathway to decertification for low performing OPOs.25 The new rule is a big step forward, and we strongly urge HHS to apply the new measures to the upcoming 2022 certification cycle to enact much-needed change as soon as possible.26 The new rule also highlights just how dire the current OPO situation is: 34 out of the 57 OPOs in the country are so sufficiently underperforming that they would either be decertified or their donation service area would be opened up for competition.27 28
For the many reasons discussed so far, it is essential to identify the oversight gaps that have allowed such egregious OPO misspending, underperformance, and patient safety issues to go unremediated. As one transplant center consultant explained, “I don’t think anything on the OPO side will improve until they feel like they can’t get away with hiding things.”
Our research revealed oversight gaps or conflicts in the following 6 key areas:
- OPO failure to recover enough organs, quality control, and negligence
- Complaints process
- Transparency around conflicts of interest
- Communication across a diffuse government structure
- Financial incentives
- Technology and security
The purpose of this report is to closely examine the current governance and oversight structure of the organ donation system to identify gaps, conflicts, and impotencies. We also provide recommendations for what can be done to save more lives through organ donation and transplantation. Organ donation reform is more urgent now than ever before, given evidence that COVID-19 increases the risk of organ failure and dialysis centers pose a major transmission risk to vulnerable patients.29 30
While many of the entities and regulations in this report also pertain to transplant centers, we will be focusing primarily on the oversight of OPOs, given their monopoly government contractor status, clear documentation of their severe poor performance in organ recovery, and their financial opacity, as well as the failure of OPTN/UNOS to properly oversee them. Additionally, the oversight system for transplant centers appears to be working more effectively,31 while there is clear evidence that the oversight system for OPOs and UNOS is broken and in urgent need of addressing.
Current Reliance on Media for Accountability
Many failures within the organ donation system have either been largely ignored by regulatory bodies, or weren’t given attention until after they garnered press. As one government official told us about the organ donation system, “sometimes the best way to file a complaint is to go to the news media.” This was the path Oregon resident Erika Zak and her family took after a policy change made it more difficult for her to receive the liver she needed.32 UNOS, the federal contractor tasked with overseeing OPOs, made the policy change that resulted in a lower “score” for Erika’s placement on the waitlist. The change led to a score that was “determined not by her doctors’ careful evaluations, but by an anonymous panel of five transplant professionals who reviewed a short application,” according to the New York Times.33 As Erika’s twin sister, Jenna, told us, “Erika and [her husband] Scott were cautious about speaking to the press, but at the time they felt like they had no other options than to call out UNOS.” Before going to the press, Erika and her family wrote letters, reached out to UNOS directly, reapplied for a new score, and had her doctors argue her case. But UNOS stayed quiet, according to Jenna. The effort of taking their plight to the media “felt like a full time job,” and was a drain on their family who was already going through so much. Jenna told us that, “[s]omeone said UNOS’ strategy was just to sit it out…thinking that we would give up or that Erika would die.” Erika’s story was covered by outlets such as CNN and the Washington Post. The New York Times editorial board even profiled her case, concluding that HHS should “fix the UNOS scoring system” and “revisit the UNOS monopoly” to help address the “astounding lack of accountability and oversight in the nation’s creaking, monopolistic organ transplant system [that] is allowing hundreds of thousands of potential organ donations to fall through the cracks.”34
“Sometimes the best way to file a complaint [about the organ donation system] is to go to the news media.”
- Government official
While Erika’s story received national attention, there are thousands of organs that are lost and patients who subsequently die and never make it to the press. Relying on the media to catalyze change in this system is both insufficient and unfair. This approach leads to a reactive system of oversight, addressed by a patchwork of fixes.35 And it exacerbates inequity issues within the system. As Erika’s sister told us, “we were in a position where we were privileged to have those [media] connections. Other people don’t have those opportunities, even though they’re struggling with the same situations… it’s completely unfair. That’s not how this should work.”
While transplant centers have historically faced a wide range of scrutiny from the press,36 37 OPOs may miss a clear opportunity to recover a life-saving organ, and it’s very possible no one outside the OPO finds out or files a complaint. The scrutiny on transplant centers has led to more public inquiries from oversight agencies,38 and a wave of regulatory changes.39 However, no such wave has happened with OPOs yet.
Every organ that is not recovered because of OPO ineffective practices, transportation errors, or understaffing, results in another person dying while on the waitlist. For the purposes of this report, we have termed these “shadow deaths”; they are just as real and consequential as deaths resulting from errors that have brought higher profile media attention, but are less visible as they result from systemic rather than acute problems. This aligns with findings from our earlier research that, in the estimation of one leading researcher, “we don’t have an adequate way of expressing the harm of a non-approached donor. There are significant harms – the donor’s decision to donate may not be honored, the family may not get closure or comfort, patients on the waiting list die, and costs increase to the national health care system. And yet OPOs are able to keep these harms invisible.”40 A large oversight gap in the organ transplant system is in accounting for these “shadow deaths,” and the 110,000 people currently on the transplant waiting list deserve immediate action to address them.
Overview of How the Governance was Designed
The Department of Health and Human Services (HHS) currently oversees the organ donation system. Within HHS, Centers for Medicare and Medicaid Services (CMS) is responsible for overseeing OPOs. Historically, the Health Resources and Services Administration (HRSA) was responsible for overseeing the OPTN, however as of publication of this report (January 2021), this role is being shifted to the Office of the Assistant Secretary for Health (OASH).41
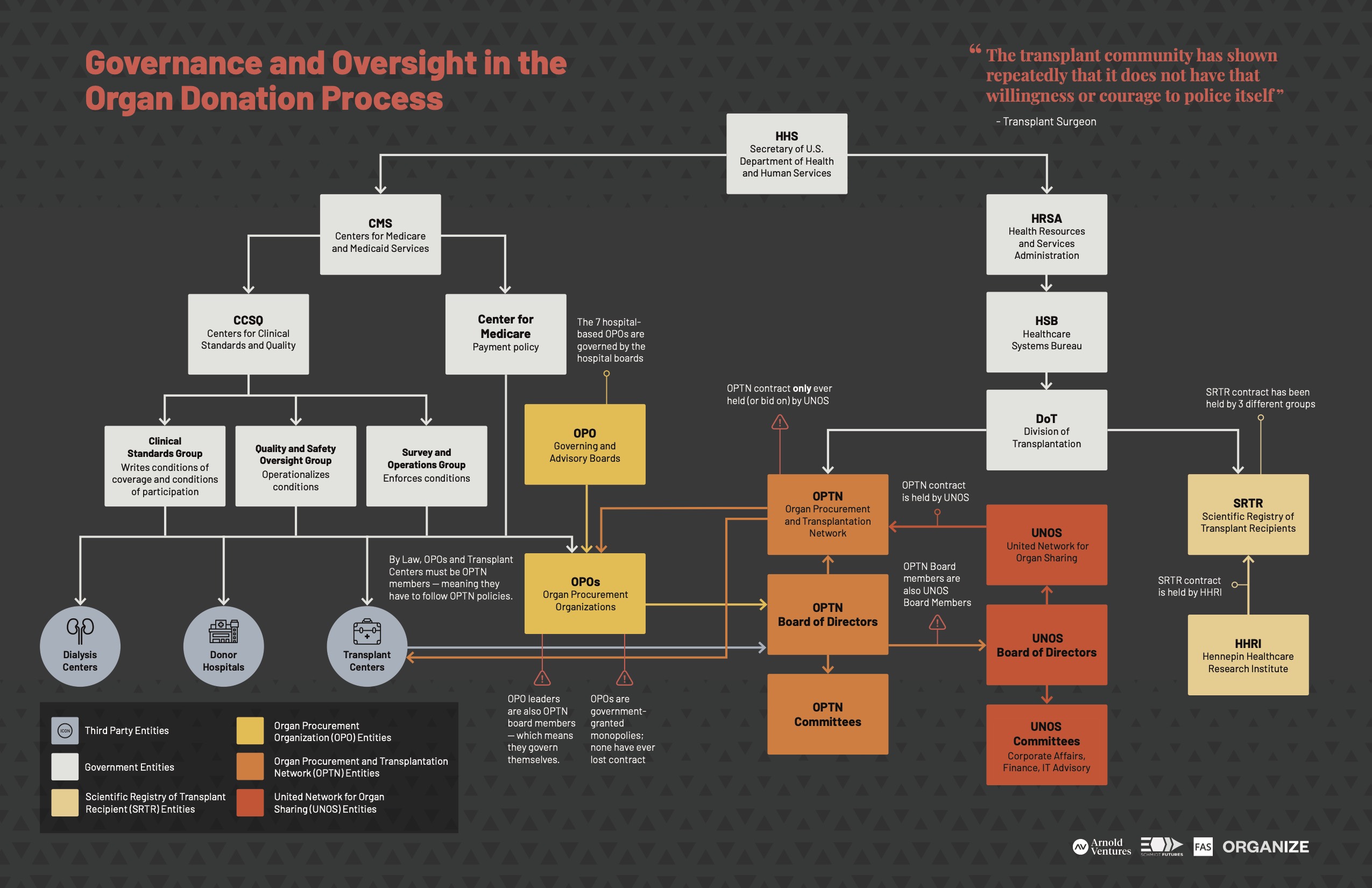
Download the “Governance and Oversight in the Organ Donation Process” PDF
CMS Oversight of OPOs
CMS is responsible for certifying OPOs and establishing the rules OPOs must follow – or the “conditions for coverage” – in order to be compensated for their organ recovery and placement work. CMS, specifically the Center for Clinical Standards and Quality (CCSQ) and the three sub-departments under CCSQ, are responsible for writing the conditions for coverage for OPOs, enforcing the conditions, and recertifying OPOs.42 The Center for Medicare (CM) within CMS pays the OPOs for organ recovery through reimbursements.
The 2020 Final Rule, which is projected to increase the recovery of life-saving organs by more than 7,000,43 notes that historically OPOs have not been held accountable for consistent low performance.44 CMS’ press release announcing the Final Rule stated, “[s]ome stakeholders have argued that many [OPOs] are underperformers and have faced few consequences for their poor performance. Current organ recovery and transplantation measure regulations are outdated and allow OPOs to subjectively report organ recovery rates.”45
As one transplant surgeon explained, “there’s been a tremendous amount of inefficiency and issues associated with the OPO system, [but it] has never crawled to the top of the CMS agenda.” Some within CMS perceive the overall cost of compensating OPOs as small compared to the total dollars spent elsewhere, and so therefore not worth much attention. As one senior government official told us, the historical lack of attention paid to OPOs is because “it’s such a small part of the total dollars that CMS uses and administers every year.” However, the relatively small reimbursement cost to OPOs shadows a much larger cost of OPO inefficiency; CMS currently pays $36 billion a year on dialysis and treatment for people who need a kidney transplant – more than the annual budget for NASA46 and the CDC47 combined.48 Each kidney transplant facilitated represents a lifetime cost savings of more than $350,000 per patient in avoided dialysis costs.49 50 Research estimates the U.S. could save $40 billion over 10 years by increasing kidney transplant via OPO reform.51
“Research estimates the U.S. could save $40 billion over 10 years by increasing kidney transplant via OPO reform.”
OPTN Oversight of OPOs
When the National Organ Transplant Act (NOTA) was passed in 1984, it established the OPTN – a membership-based entity tasked “to improve the effectiveness of the nation’s organ procurement, donation, and transplantation system by increasing the availability of and access to donor organs for patients with end-stage organ failure.”52 All OPOs are OPTN members, and thus are expected to comply with OPTN bylaws and policies.
The OPTN contractor role has been filled exclusively by UNOS since 1986. However, while the OPTN contract is awarded by the federal government, more than 90% of the contractor’s revenue comes via stakeholders who are required by the government to be OPTN members, seemingly creating an inherent conflict; investigative reporting from the Los Angeles Times covered this dynamic, describing UNOS as a “reluctant enforcer” and noting that “[s]uch collegiality is built into UNOS’ very structure – and that’s the problem, some critics say. UNOS isn’t just a regulator; it is a membership organization, run mostly by transplant professionals.”53
Many people we spoke with reinforced this characterization, suggesting that UNOS seems more concerned with keeping their members (OPOs and transplant centers) happy than exercising legitimate oversight; in effect, shielding its members from punishment rather than actually holding them accountable.
But this need not be the case. NOTA tasks the OPTN (currently UNOS) with improving the effectiveness of the organ donation system, and gives UNOS plenty of leeway to oversee all aspects of OPO practices. Specifically, NOTA requires the OPTN to “work actively to increase the supply of transplantable organs,”54 and according to sources within the government, UNOS’ contract also explicitly charges them to “identify root causes of issues” pertaining to OPTN members, including OPOs.
The fundamental problem seems to be that UNOS, as the sole OPTN contractor, has chosen to deny responsibility for several aspects of OPO oversight.55 For example, in response to a Senate Finance Committee (SFC) letter asking what steps UNOS has taken to address the multiple OPOs who expensed taxpayers with “unallowable expenditures,” UNOS wrote that “[t]he OPTN is not authorized to monitor or enforce CMS Conditions for Coverage [and that] UNOS and the OPTN do not require independent audits of OPOs for costs because cost reporting is not an OPTN Obligation.”56
While this may be technically true, there is certainly nothing in NOTA that would preclude UNOS from overseeing such issues. In fact, understanding OPO resource allocation would seemingly fall entirely in line with UNOS’ contractual mandate to identify the root causes of OPO performance issues. UNOS’ denial of these responsibilities is clearly a choice in how they interpret the mandate given to the OPTN, and they could be doing much more to address root issues and increase the supply of organs.57
In addition to the denial of responsibility for certain aspects of OPO oversight, UNOS also does not provide any incentive for OPOs to change. Even in the exceedingly rare instances when the OPTN determines that an OPO has violated policies, the strongest punishment that UNOS metes is to deem the OPO a “member not in good standing,” a designation UNOS has only used twice in its decades-long history.58 As one HHS official told us, “the OPTN would not take strong action against an OPO unless it was doing something unthinkably egregious, like if they were buying and selling organs.” Alarmingly, documented issues such as criminal activity of OPO executives and patient safety issues that led to death have not led to OPOs being classed as “members not in good standing.”
“The strongest punishment that UNOS metes is to deem the OPO a “member not in good standing,” a designation UNOS has only used twice in its decades-long history.”
Part of the reason for this lack of enforcement is the rampant conflict of interests within the OPTN committees that are supposed to be monitoring OPO conduct. Within the OPTN, two key committees are tasked with improving OPO and other members’ performance: the Membership and Professional Standards Committee (MPSC) and the Operations and Safety Committee (OSC).
The MPSC serves to “maintain membership criteria and monitor OPTN member compliance with OPTN membership criteria, OPTN bylaws and policies, and the OPTN Final Rule.”59 And while the MPSC, in its entirety, includes a diverse set of stakeholders across the transplant community, complaints about OPOs are first routed through smaller subsets of the MPSC to which OPO professionals are preferentially assigned. This structure seems to undercut the spirit of the Final Rule, as well as to insulate OPOs from meaningful external oversight over OPOs.60
One MPSC member we spoke with said, “my observation about participating on the MPSC is that the UNOS staff seem to persuade the committee members into taking specific actions based on how previous committee members have historically treated similar issues. Precedent seems to be the most important consideration,” and that “I will not pretend that the identity of the OPO and how well one knows the people at an OPO doesn’t influence how serious an event is perceived.”
Additionally, many of the people on these committees, and on governing boards at large, represent OPOs that are failing, according to metrics in HHS’ November 2020 Final Rule.61 62 This means that staff from underperforming OPOs are creating their own performance review process, bringing into question just how effective the review process is designed to be.63 Additionally, while the OPTN and MPSC technically employ a conflict of interest policy, their chosen definitions for such conflicts are overly narrow, likely missing many actual conflicts.64 Compounding the problem, MPSC proceedings are protected by peer review, so there is no transparency around whether OPO leaders are using the process for personal or professional gain or advancement, or the extent to which cronyism protects OPOs favored by the MPSC.
The OSC, which is responsible for “improv[ing] the quality, safety, and efficiency of the organ donation and transplantation system,”65 creates suggested practices for all members to improve performance, but these are largely voluntary and do not stand alone as compliance measures. As one former MPSC chair informed us, the MPSC “never spoke about operations stuff” nor discussed issues at a systems level. These two committees do not collaborate to address systemic problems by identifying where OPO failings are a trend, but instead focus only on one-off incidents. In other words, massive underperformance – and therefore “shadow deaths” from organs not recovered – does not seem to rise to consideration of the MPSC, despite organ recovery being the central function of OPOs.
Tellingly, even UNOS appears to have previously acknowledged its role in providing oversight. As of February 2020, in the FAQ section of UNOS’ own website UNOS stated that it “manages the first two [of three] steps” of “OPO oversight.” Only after the Senate Finance Committee opened an investigation into UNOS’ abdication of such oversight did UNOS change the language on its website to distance itself from oversight responsibilities, stating: “Many people think UNOS oversees every facet of the transplant process. We don’t…UNOS is a forum for organ donation and transplant professionals to come together and determine how the national system should work.”66 According to one MPSC member, “[if UNOS actually wanted to], they could put their hands on OPO failures much more seriously than they have in the past.”
In any case, one of two things must be true: either the OPTN is responsible for such oversight and has been delinquent in exercising its authorities, in which case HHS should consider UNOS’ track record in future contracting cycles; or HHS can determine that the OPTN is not responsible for such oversight, and, by logical extension, should then reabsorb such functions into an Office of Organ Policy to ensure that OPOs are meaningfully regulated on behalf of patients and taxpayers. We note however, that in the first instance, regulatory capture could remain a persistent concern.67
Similarly, we have heard from stakeholders that the executives, staff, or analysts from the Scientific Registry for Transplant Recipients (SRTR) are allowed to engage in private consulting work for individual OPOs. This is a problem, because SRTR is supposed to be providing unbiased policy research to HHS regarding OPO performance. If executives within SRTR are viewing OPOs as potential clients, they might have the potential to distort their findings. According to one longtime OPO executive, “I had no idea about this until recently, and I was floored! It also amazes me that AOPO [the Association of Organ Procurement Organizations] consults with the SRTR about OPO performance data and the OPO regulation, and that the SRTR was actively assisting in discrediting CMS’ proposed OPO rule. In effect, a HRSA contractor was trying to help AOPO undermine CMS.”
HRSA’s Historical Oversight of the OPTN and UNOS
As mentioned above, HRSA is currently phasing out of its role in overseeing the OPTN. The Division of Transplantation (DoT), which previously lived under HRSA and will now live under Assistant Secretary for Health (OASH), is tasked with contracting out the role of the OPTN and overseeing its function.
One problem we heard from multiple interviewees is that NOTA – the 1984 law that created the OPTN – has been very narrowly interpreted. Since it was established, NOTA has been interpreted to require the OPTN be operated by a single, private contractor. This interpretation of the law, while up for debate, has been a major blocker in instilling unbiased oversight, as well as serving to suppress meaningful competition for the OPTN contract.68
According to one government official we spoke with, HRSA historically tried to break apart the OPTN contract so one expert contractor could manage the technology portion, while another contractor managed policy-making, and another managed member evaluation. This would theoretically reduce monopolism, anti-competitive behavior, and conflicts of interest, while allowing for more expertise to be used for the very different functions. Research has identified major technological shortcomings in the current OPTN contractor’s software, showing an urgent need to seek out a better vendor solution.69 However, these historical attempts to find flexibility had been met with resistance, according to our interviewees, although the incoming Administration should consider ways to increase flexibility in order to better serve patients.
HRSA lives within HHS. As one senior government official explained, another issue was that HRSA had little influence compared to other agencies in HHS – which, as they explained, leads to pressure for them to “not rock the boat” or upset the larger agencies. This also made it difficult to practice effective governance over the OPTN. HRSA’s role (and now OASH’s going forward), as the main government representative in OPTN committee meetings, is to ensure the obligations of the OPTN contract are being fulfilled. However, we were told by several interviewees that HRSA representatives often lacked the expertise to discern whether what was being said and decided was appropriate.
OPO Board of Directors
Even in the context of a breakdown in governmental oversight, an OPO’s board of directors, in line with their fiduciary responsibilities, can still wield considerable influence on OPO performance. However, many stakeholders we talked to suggested that many OPO boards are plagued by ineffectiveness, conflicts of interest, and inconsistent governance. “[OPO leadership] are not accountable to anybody but the people they choose to be on their boards,” explained one former government official, “so it serves them to have people on their board who are their buddies.”
“[OPO executives] are not accountable to anybody but the people they choose to be on their boards, so it serves them to have people on their board who are their buddies.”
- Former government official
Some OPOs further incentivize their board of directors to cooperate by paying them for various services.70 For example, OneLegacy, the Los Angeles OPO, which under the November 2020 outcome measures was flagged as tier 3 “failing,”71 nevertheless pays its board chair, Bill Chertok, $100,000 and other board members $30,000 or more.72 Despite being one of the worst performing OPOs, as well as having been found by the HHS Inspector General to profligately misspend taxpayer funds, it appears the CEO is insulated from being replaced because of such payments.73 Given that OPOs are essentially reimbursed 100% through Standard Acquisition Charges (SAC fees) and CMS reimbursements,74 it is not a cost to the CEO of OPOs to pay board members, but rather a cost to taxpayers. Additionally, while OPOs are technically not allowed to receive reimbursement for lobbying expenses, board compensation creates a loophole. For example, an OPO can compensate a politically-connected board member and leverage such access, which would not technically qualify as lobbying.75
While NOTA prescribes the composition of OPO advisory boards – which include clinicians, such as transplant surgeons and hospital representatives – there are currently no regulations for who is on an OPO governing board. The advisory boards make general recommendations to the board, but it is the governing board that decides whether or not to act on them. One HHS official shared that there have been high-level discussions about the need for more regulations around who is on the OPO governing board, and agreed that there should be requirements around qualifications and type of representation. However, it is uncertain what authorities HHS has to direct the composition of the OPO governing board, which has led to highly inconsistent roles, policies, functions, and effectiveness of OPO governing boards, allowing poor OPO performance – and rampant “shadow deaths” – to persist. 76
Even when an OPO board of directors means well and has the right qualifications, it is often limited by the data that they’re shown by the OPO. One OPO board member we spoke with said the lack of shared data regarding OPO performance hampers the ability for general improvement. “The OPO board is only as good as the information they get… The evaluation tools that we get to show us how we’re doing compared to other OPOs aren’t helpful. They all give us a false sense of security that everything’s fine. A lot of OPO boards don’t get comparative data, they don’t get to see what their peers are doing.” One OPO CEO who helped turn around an underperforming OPO in less than a year explained that one board member’s response was: “We have to let everyone know that the OPOs are broken. I personally feel duped that I’ve been told all these years [that] we were doing everything we could.” This problem is exacerbated by UNOS and OPOs, which regularly herald record organ donation rates, despite peer-reviewed research showing that this is due to the opioid epidemic and not OPO improvement.77 78
Diffuse Governance and Why That’s a Problem
Multiple people we spoke with pointed to the lack of coordination and conflicting tensions between the various arms of oversight as being one of the largest problems within organ donation. “Governance of organ donation is remarkably diffuse and not terribly well coordinated,” explained one HHS official. Others characterized organ donation oversight as too big for HRSA and too small for CMS. While this may be perceived as a relatively small cost for CMS when only considering direct OPO reimbursement, it is a massive expense when we consider the dialysis costs79 stemming from OPO failure to recover enough kidneys for patients who could have been transplanted but were not. Thus this lack of effective oversight is not only costing lives but also taxpayer money.
Oversight coordination has become a topic of congressional interest, as a recent letter from the Senate Finance Committee to HHS requested: “Please provide all documentation regarding coordination between HRSA, CMS and the OPTN contractor (currently UNOS) related to the following three cases involving lapses in patient safety.”80
CMS handles the money and ultimately determines whether an OPO is certified (and thereby eligible for CMS reimbursement for services). Pertaining to deceased donation, the Department of Transplantation (DoT) (formerly under HRSA and going forward under OASH) is charged with awarding and managing the OPTN contract, and has indirect influence over the waiting list and organ allocation. But the lack of clear delineation of responsibilities between oversight authorities (HRSA and CMS as agencies and UNOS as the current contractor operating the OPTN) has historically led to many inefficiencies in an ecosystem that would benefit from more streamlined and holistic policymaking. “It is unclear as to what the individual responsibilities are. And that’s the kernel of the issue,” said one UNOS board member.
“It is unclear as to what the individual responsibilities are. And that’s the kernel of the issue.”
- UNOS Board Member
The “quasi-governmental” nature of the OPTN has also opened questions about which entity is accountable in various and all-too-commonly occuring scenarios: the OPTN (currently UNOS), HRSA, or CMS. The path to accountability has been further muddied by the sweeping language within NOTA, and the discrepancies in interpretation across the OPTN.
Gaps and Conflicts in Oversight
Key areas of oversight gaps we identified include: OPO poor performance and negligence, the reporting process for complaints and misconduct, transparency about (and lack of meaningful prohibitions against) conflicts of interest, cross-governmental communication, financial incentives, and technology and security.
We share these findings with the intention of shedding light on the aspects of the organ donation system that need better accountability and oversight, with the ultimate goal of recovering more organs and saving more lives. We are aware that some stakeholders within the system have argued that highlighting these issues may undermine public trust in the donation process and lead to fewer donations, but historically such attention simply has not had any depressing impact on donation rates. For example, even in an extreme case of bad press regarding a transplant surgeon charged with felony murder in 2008 for allegedly hastening someone’s death in order to recover transplantable organs, the Los Angeles Times found that the case had “not dampened organ donations.” And that according to a spokesperson for the Los Angeles OPO, “consent rates at hospitals go up and up.”81
In reality, media spotlight on shortcomings in the system has been a crucial tool in effecting change. Members of Congress,82 83 including those in the Senate Finance Committee and the House Committee on Oversight and Reform,84 have cited several investigative journalism pieces as evidence of why OPOs need to be held to higher standards.85 86 87 The Congressional letters boosted by the media pieces helped usher in the Final Rule, which is projected to lead to thousands more organs recovered, and thus save thousands of lives. Research suggests that heightened scrutiny of OPOs – resulting from increased awareness of OPO failures – actually likely has a positive effect on donation rates. A report from the Bridgespan Group, citing data from leading researchers in a public comment to HHS regarding the then-proposed OPO rule, found: “Since the executive order announcing the proposed new metrics and increased oversight, data show that OPO performance has already begun to improve, perhaps early evidence of the ‘Hawthorne effect’ (i.e., increased scrutiny and observation by itself drives behavior change that leads to improved outcomes).”88 89 In fact, according to patient advocates, when legislators assume goodwill within the organ donation system, it can block further scrutiny and stall reforms that would strengthen the system and save more lives. 90
OPO Failure to Recover Enough Organs and Negligence
OPO failure to recover enough organs and negligence is a key area that reflects major gaps in oversight. As mentioned earlier, OPO performance varies widely. Until the November 2020 Final Rule, OPO regulation relied upon self-interpreted, self-reported data, which made it easy to manipulate. As one OPO leader told the New York Times, “I used to work at an OPO, and we reported false numbers to make it appear we were doing better than we were.”91
Circularly, such data reporting issues have also made it impossible for the government to actually decertify OPOs or otherwise hold them accountable.92 The Association of Organ Procurement Organizations (AOPO), the trade group representing OPOs, has argued that because data is self-reported and unverifiable, “the accuracy and consistency of data cannot be assured.”93 This is the same argument that prevented CMS from decertifying the Arkansas OPO in 1999, despite the OPO failing four out of five performance measures then in use; a federal judge agreed with the OPO’s case that CMS could not rely on the OPO’s faulty data as the legal basis for decertification.94
Our research also revealed several additional problems with current OPO accountability measures, specifically the following:
- Ineffective site surveys
- Unregulated and ineffective transportation of organs
- Absence of process metrics and data
- Understaffing
Ineffective Site Surveys
Aside from their self-reported data, the primary ways OPOs are evaluated come from infrequent site surveys conducted by UNOS, CMS, and – optionally – the Association of OPOs (AOPO). The UNOS site surveys do not measure how well an OPO performs at recovering organs, but rather how compliant an OPO is with OPTN policies. Given that, as outlined above, the OPTN is greatly influenced by its OPO members, this process can become more of a checkbox for UNOS to go through the motions of an evaluation rather than any true scrutiny and oversight of OPO conduct.
UNOS only reviews data from OPO’s electronic health records (EHRs) and compares it to DonorNet (the OPTN technology that matches organs with patients on the waiting list). In their audit, UNOS does not cross reference DonorNet data with donor hospital records to ensure the information is correct and accurately reflects the OPO’s performance. This is a huge gap in ensuring accurate reporting by OPOs. OPOs can easily manipulate the data by only entering information into their system that casts them in a favorable light. For example, if an OPO was not able to respond to a potential donor referral in time because of understaffing, they could leave the patient record completely out of their EHR records or put another reason as to why the referral did not become a donor. In effect, UNOS is simply auditing whether the OPO’s reporting is internally consistent (even if incomplete or misleading), rather than actually assessing the OPO’s performance.
“UNOS audit is a joke. All that the UNOS auditors do are come in and say, ‘do you meet the policies and requirements that are set forth, yes or no.’ They are not there to improve things…There’s nothing that tries to push you further.”
- OPO consultant
As one OPO consultant explained, “UNOS audit is a joke. All that the UNOS auditors do are come in and say, ‘do you meet the policies and requirements that are set forth, yes or no.’ They are not there to improve things…There’s nothing that tries to push you further.” Another OPO leader shared, “[the site survey] is not a survey that will tell you how good an OPO we are. It will tell you how compliant we are with the administrative and clinical policies that they [the OPTN/UNOS] have.”
Governmental surveys are likewise fraught. Several people we talked to said CMS surveys were conducted by surveyors without proper subject matter expertise to truly assess the organ donation process. As one OPO leader said, “these are the same surveyors as those inspecting meat packing plants.” Even surveyors with medical facility expertise would miss many of the problems of an OPO operation because OPOs do not function in the same way as hospitals and other medical providers. Other people we spoke to said the CMS survey required a lot of paperwork, but that it was easy to “spin it” to the auditors.95 As another HHS official shared, “it’s clear that CMS doesn’t have the expertise to be able to have oversight [of OPOs] effectively… We need surveyors who understand the nuances, and can identify problems more effectively.”
Most OPOs volunteer to participate in a third optional survey by AOPO, to get accredited as an AOPO member. But since AOPO is not an oversight body, findings from such surveys do not lead to any consequences for an underperforming OPO. Some OPOs find these surveys to be more helpful because it has fewer consequences from an oversight perspective. As an OPO leader shared, “when AOPO was coming in, I always told staff ‘be honest’ because what you say to them is not going to change our being funded, it’s not CMS. We’re choosing to be accredited, so if there is something that would drive additional staffing, supplies, or technology resources that’s needed, this could come out of the AOPO survey. CMS is not the place you encourage staff to say, ‘yeah, we’re understaffed and we can’t keep up the call schedule,’ because of course everyone worries you’ll be slapped on the hand and lose funding.” However as an OPO executive informed us, AOPO surveyors are staff from other OPOs, some of which are low-performing OPOs or may be friends of the OPO they’re surveying, so there is often an inconsistent relationship between the standards and how effectively the OPO is performing. While they may provide some value, optional and consequence-less surveys from the OPO trade association should not be confused with, or used to crowd out, necessary oversight.
Unregulated Transporting of Organs
There are also troubling gaps in what surveys examine. One of the many things that the site surveys don’t look at is how well organs are being transported to where they need to be for transplantation. Organs lost or delayed in transit directly lead to patients dying on the waitlist or suffering worse clinical outcomes due to increased cold ischemia time of the organ.
Evidence shows that transportation failures are a common occurrence. For instance, a recent Kaiser Health News investigation found that hundreds of organs have been lost, damaged, or delayed in transit, “often rendering [the organs] unusable…[and that this happens because organs] are typically tracked with a primitive system of phone calls and paper manifests, with no GPS or other electronic tracking required.”96 Based on the numbers reported, organs appear to be getting delayed or lost on a weekly basis.97 Both organs transported through the OPO, as well as those by the UNOS Organ Center, have experienced significant delays, damage, and costs. Despite CMS conditions for coverage for OPOs that explicitly state that, “[t]he OPO must develop and follow a written protocol for packaging, labeling, handling, and shipping organs in a manner that ensures their arrival without compromise to the quality of the organ,”98 to our knowledge no oversight investigation has been performed by CMS resulting from Kaiser’s reporting or subsequent Congressional inquiries,99 and the OPOs responsible still have not experienced any repercussions from CMS.
The OPTN, whose role is to promote “efficient management of the [organ donation] system,”100 has no standard policy around transportation, so each OPO does it their own way. This is despite an explicit obligation in UNOS’ contract to “provide technical assistance to OPOs, 24 hours/day, every day of the year, in organ placement to facilitate matching, transporting, and sharing of organs, and to monitor and reduce organ discards resulting from logistical issues in the system.”101 In a seeming abdication of its mandate to assist and monitor OPOs in this process, when questioned about its role in organ transportation by the Senate Finance Committee, UNOS responded that “the OPTN does not collect transportation data on a national, systematic basis” and that “[m]atters involving the transportation methods used by organ procurement organizations (OPOs) are arranged directly between OPOs and transplant centers.”102
It should also be noted that UNOS directly handles a subset of organ transportation cases via the UNOS Organ Center, the deficiencies of which were reported on in the Kaiser Health News expose. When the Senate Finance Committee asked UNOS about the findings highlighted in the expose, UNOS stated that they had only started recording data for organ transport handled by the UNOS Organ Center as recently as 2016.
UNOS responded that 409 organs transported by the UNOS Organ Center have experienced transportation issues since 2016, and of those, 133 organs were never transplanted.103 This means that UNOS itself, as the OPTN contractor that is supposed to oversee OPOs, is responsible for hundreds of organs being transported in ways that have led to delays and damages, ultimately resulting in more than 130 “shadow deaths”104 of patients who were never able to receive those organs.
Importantly, as UNOS also noted, these transportation cases handled by the UNOS Organ Center represent only a “small subset of organ transportation arrangements” and that the “vast majority of organ transportation arrangements are not facilitated by the UNOS Organ Center.” By extension, it is reasonable to assume that the real total of organ transportation failures are likely orders of magnitude larger than what Kaiser Health News was able to report; however, because of UNOS’ failure to capture and report complete and transparent data, we still do not know the exact number of organs that incur damage due to transportation issues across all OPOs. We also don’t know the full extent of harm caused to patients as a result, including the total life-years lost to patients because organs were never transplanted or because the quality of an organ was degraded as a result of a travel delay.105
In addition to the transportation cases covered by Kaiser Health News, several transplant surgeons we spoke with described severe problems with organs in transit, including:
- Receiving a frozen kidney, packaged incorrectly by OPO106
- Receiving an damaged organ with tire marks on the box
- Failing to receive a pancreas that was locked in an airport locker and never picked up by a courier
Each of these instances results in a patient’s death, given that one fewer organ is used for transplant. And yet, so far as we could tell, none led to investigation or action by UNOS despite physicians alerting UNOS to the problems. Whether physicians also alerted CMS in these instances is unclear; which is itself telling that there is not a belief that the government will act.
Absence of Process Metric and Data
One of the biggest gaps with the OPO oversight is that HHS does not evaluate key process metrics, such as: hospital referral trigger criteria; triaged referral responses by OPO; response time to referral; adherence to donor management guidelines; discard rates; organ acceptance rate; consent rates; adherence to consent process; consent not recovered; or adherence to organ/tissue labeling. According to a former CMS official, “there are no real process measures that measure their effectiveness. And [those process measures are] all over the map depending on the OPO.”
The metrics evaluated by CMS are all for outcomes, which are currently fraught for reasons discussed earlier, and even HHS’ new Final Rule for OPOs does not take effect until the next OPO contracting cycle in 2022 (with failing OPOs possibly not being decertified until 2026). Not only is this bad for evaluating and overseeing OPO performance, but it also gives OPO a false sense of confidence in their performance when the data is not aggregated and shown in the context of all 57 OPOs. As one OPO CEO told us, “the number one thing is, we think we’re doing fine. That’s the irony of all this. All of the evaluation tools that we have from CMS, OPTN, tell us we’re doing fine…. Nobody in an oversight capacity, nobody at CMS, no one at HRSA, no one at HHS knows enough to tell us anything differently…That’s the big problem.” One transplant surgeon reinforced this assessment, noting that “there’s a big gap between the organs available and where we start reporting.”
“The number one thing is, we think we’re doing fine. That’s the irony of all this. All of the evaluation tools that we have from CMS, OPTN, tell us we’re doing fine…. Nobody in an oversight capacity, nobody at CMS, no one at HRSA, no one at HHS knows enough to tell us anything differently…That’s the big problem.”
- OPO CEO
Data on these process metrics are needed to establish industry standards for OPOs to measure against and gain a truer understanding of their performance. This can also help OPOs better understand some of the root causes driving poor outcomes, especially as UNOS has seemingly eschewed this responsibility.
OPO Understaffing
One of the root causes identified for OPO failure to recover organs is chronic understaffing. As one OPO leader told us, “OPOs really need to stay overstaffed so that they can meet the demands when there’s an influx of potential donor referrals, but they don’t like to do this because of cost.” (Note: since OPOs are 100% reimbursed for costs,107 OPO understaffing more often results from OPO misallocation of resources rather than OPO resource-constraint.)
While there is regulatory language around staffing from both CMS and the OPTN, functionally, neither entity closely examines nor addresses the issue of understaffing. Data on OPO staffing has long been a subject of research interest, though neither AOPO nor individual OPOs have volunteered such information, nor has HHS mandated its disclosure. While CMS requires in its Conditions for Coverage that “OPOs must have a sufficient number of qualified staff,”108 this language is very broad and does not specify how to evaluate what is considered ‘sufficient’ staff, nor does it address the chronic issue of OPOs not having enough staff to meet the peak needs when they occur. There is also no indication that CMS has initiated any investigations in response to reports of an OPO being “short-staffed at critical moments.”109
UNOS similarly does not identify understaffing as a systemic issue that results in organs not being procured. When asked about it by the Senate Finance Committee, UNOS cited that there is language in the OPTN bylaws that, “Each OPO must have the necessary staff to recover and distribute organs according to OPTN obligations” but functionally, all that they require is for OPOs to notify the OPTN when their administrative director or medical director changes.110 Supposedly, UNOS will initiate a patient safety/non-routine compliance review if they learn of staffing shortages, but there is no evidence that any OPO has experienced repercussions from UNOS for staffing storages. Even in response to a direct question from the Senate Finance Committee on how UNOS responded to a media report about an understaffed OPO that resulted in the failure to recover organs, UNOS could not provide any information.111
One former OPO leader we spoke with recalled a time when a 40-year-old candidate for organ donation passed away and his organs were never recovered. “It just boiled down to staffing. Someone had worked 26 hours, and no one was there to recover the organs. [The OPO should have] made sure there was staff there for every opportunity.” Cases like the 40-year-old candidate not getting recovered are often either categorized as a “missed opportunity” or not reported at all. But as an OPO leader explained, it’s more than a missed opportunity, “it means more people died overnight that could be alive right now… just because we didn’t read the obituary doesn’t mean someone didn’t die.”
Not only are OPOs understaffed, but the staff they do employ also does not appropriately reflect the communities they serve. Studies have found that donation rates are higher when OPO staff discussing organ donation with donor families are the same ethnicity and speak the same language,112 113 yet OPO staff remain overwhelmingly white114 and often speak only English.115 This impacts donation rates, which in turn lowers the chances of people of color finding a suitable match – as recipients are most likely to match with a donor of the same ethnicity.116
Process for Reporting Complaints and Misconducts
In addition to oversight gaps in OPO underperformance, we investigated several avenues for how people can lodge a complaint when there is OPO or OPTN misconduct. We learned that complaints are actually seldom officially lodged against OPOs or UNOS, generally for three main reasons:
- Fear of retribution if someone makes their concerns known
- Lack of confidence in the system that a complaint will lead to change
- Unawareness of the reporting process for a complaint
One transplant consultant explained to us, “if I had a complaint, I probably wouldn’t go anywhere. The messenger usually gets shot, so knowing what I know, I wouldn’t go to UNOS or CMS.” Several interviewees were unaware of how they would even start the complaint process, though many said they never looked deeply into it because they had so little confidence that anything would change even if they did complain. When we asked a transplant surgeon where he’d go to file a complaint about some of the problems he had seen, he said, “I honestly don’t know, because I have never gotten that far.”
One interviewee we spoke with witnessed UNOS’ Organ Center dangerously advise an OPO to misclassify an organ with a communicable disease. “I saw in real time suboptimal performance from the [UNOS] Organ Center that could have endangered patients…The gaps in what the contractor has provided to the OPOs are so apparent. They don’t know how to do their jobs on a very basic level to recover a deceased donor with an additional comorbidity…I really should have reported them to the MPSC, but they [UNOS] are the MPSC.” Instead of taking steps to address its dispensing of improper — and likely dangerous — advice, UNOS ended up investigating the interviewee and the OPO for going against the UNOS Organ Center’s guidance and placing an “out of sequence” organ. The interviewee said their employer ended up getting investigated by UNOS and explained that “those guys made our life hell.”
“A big reason no one reports anybody is fear of retaliation,” said a transplant center consultant. Researchers we spoke with mentioned taking great pains to avoid getting on UNOS’ or the OPOs’ “bad side” since they rely on data access and “letters of support” from UNOS and OPOs to apply for grants. Several researchers suggested UNOS made it exceptionally difficult to receive data requests if they knew the seeker was a researcher who was critical of UNOS. Peer reviewers for research papers are also often in UNOS or OPO leadership positions and may discredit research that is unfavorable to UNOS or OPOs. This results in researchers not feeling empowered to find and share truths that could help improve the organ donation field as a whole. Given that most policy is informed by peer-reviewed research, the stifling of research that challenges the status quo serves to further ossify the current, unaccountable system. Alarmingly, some transplant surgeons have also voiced concern that, if they speak out against UNOS, their patients would be disfavored in future UNOS organ allocation policies. Whether or not this concern is well-founded, it is telling that such an environment has been created in which this belief would be held and that it serves to chill dissenting opinions.
Even though many complaints are never made for the reasons explained above, formal complaint processes do exist through UNOS and CMS. However, the effectiveness of these processes are highly questionable given the dearth of substantial change that has resulted from either process. If an OPO experiences a patient safety issue or deviates from an established policy, it is expected to self-report the issue to UNOS through the Improving Patient Safety (IPS) portal.117
As we heard from OPO leaders, these deviations may include:
- a “miss” – when a procured organ does not make it to a transplant surgeon while it is still viable;
- a “near miss” – when transportation delays cause an organ to nearly miss the window of a viable transplant;
- And an “out of sequence placement” – if an OPO bypasses the UNOS matching algorithm and places an organ to someone further down the list.
“There really is no incentive, other than the integrity of this organization, to self report. Unless it’s something pretty egregious or if a center reports us, I’m confident there are a lot of OPOs that don’t self report that much…[UNOS] doesn’t do anything to really encourage transparency in the system.”
- OPO leader
OPOs are expected to submit their reports explaining what happened. However, as even UNOS admitted, “[s]ince reporting is voluntary, [it] is thus subject to underreporting.”118 As one OPO leader said, “there really is no incentive, other than the integrity of this organization, to self report. Unless it’s something pretty egregious or if a center reports us, I’m confident there are a lot of OPOs that don’t self report that much…[UNOS] doesn’t do anything to really encourage transparency in the system.” One OPO leader expressed that OPOs are disincentivized to self report, because they have a “permanent record” with the OPTN that is never expunged, even if they fix the issue. As an organization tasked with optimizing the network of organ donation, it is troubling that UNOS is not capturing critical data on misconduct more accurately, when those actions contribute to thousands of shadow deaths each year.
UNOS’ patient safety department reviews OPO self-reported submissions, along with any complaints from the public through emails, calls, or media coverage and decides whether or not to send them to the MPSC to review. The MPSC can then decide whether or not to conduct a peer-review trial investigation and move through steps to take corrective action. (See OPTN Complaint Map.) This peer-review process, including the process by which the MPSC decides which cases to review, is not open to public scrutiny.
OPTN Complaint Map
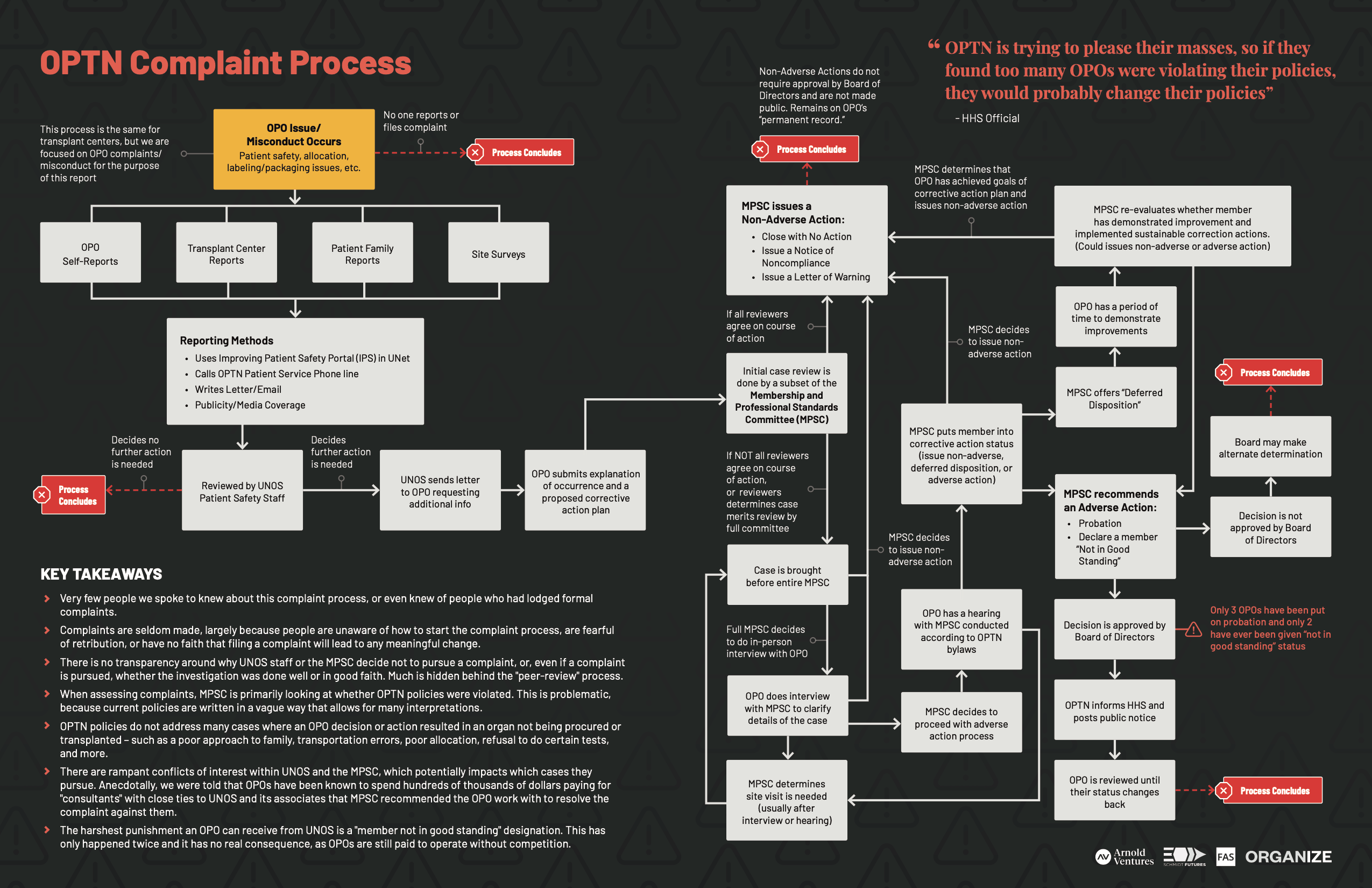
Download the “OPTN Complaint Process Map” PDF
In its response to the Senate Finance Committee inquiry about OPO misconduct, UNOS hid behind this peer-review process, citing, “we respectfully cannot provide these materials because they are privileged, confidential medical peer review information [and that] the success of our member improvement processes are critically dependent on the trust our members have in the confidentiality of this process.”119 Further review would suggest this is a logical fallacy, however, as MPSC members should not have any reasonable expectation of the confidentiality on which UNOS suggests their peer-review participation relies. This peer-review protection privilege does not extend to withholding documents from the HHS Secretary, according to the OPTN Final Rule120 and OPTN Bylaws (as of August 2020). As specified in the Bylaws, “the OPTN Contractor is required to provide the Secretary with any information acquired or produced under the OPTN Contract, including information that would otherwise be protected by the medical peer review privilege.”121
UNOS has even been forced to turn over peer-review protected information in the past after they received a subpoena from the HHS Office of Inspector General (OIG). At the time, UNOS appealed the subpoena – also on the grounds that it would undermine their future peer-review processes – though the judge ruled in favor of the OIG, writing “the inquiry is clearly within the authority of the agency [HHS OIG], the demand is clear and the information sought is reasonably relevant.”122 Going forward, Congress can use this case as precedent for future subpoenas. HHS should leverage its existing oversight authorities to access information, even absent a subpoena, and perform an audit of historical MPSC proceedings to assess the efficacy and fidelity of the MPSC process.
“Even if an OPO is deemed “not in good standing,” or put on “probation,” it is still able to function as a monopoly establishment in their designated area, and receive payment from CMS.”
Even when a misconduct is identified, the highest degree of punishment an OPTN can impose is changing a member’s status to “not in good standing,” which leads to a public announcement, but very little functional consequence. In the history of UNOS, only two OPOs have had their status changed to “member not in good standing.”123 Of those two, one had some media coverage, and the other did not. Even if an OPO is deemed “not in good standing,” or put on “probation” – the designation before being “not in good standing” – it is still able to function as a monopoly establishment in their designated area, and receive payment from CMS. OPOs found “not in good standing,” are referred to CMS (more on that below) for further investigation, but no OPO has ever been decertified as a result of this or any other complaint process. In fact, one of the OPOs that was designated “member not in good standing” went on to nearly kill a recipient of a uterine transplant because they failed to identify and flag a fungus on the uterus.124 UNOS did not disclose why the OPO initially received this designation, nor how and why it was subsequently restored to “good standing” status even despite this near-fatal patient safety lapse.125
People we spoke with said that cases that go through the MPSC generally focus on one-off failures, and that there is a missed opportunity to address the systemic issues. “I don’t think the OPTN has a means to look specifically at systems failure, and address them. The right answer is to look at a systems approach, rather than looking at individual events…One of the big flaws of the OPTN structure is that it has very little system approaches,” said one transplant surgeon.
UNOS says that their research department analyzes all submitted issues and delivers them to the OPTN Operations and Safety Committee (OSC).126 But one MPSC member we spoke with claimed the MPSC never talked to the OSC about system functions and systemic issues. The OSC has created suggested practices for all members to improve performance.127 However, these are largely voluntary. By UNOS’ own admission, “guidance from the OPTN does not carry the weight of policies or bylaws. Therefore, members will not be evaluated for compliance with this document.”128 As one person we interviewed put it, “the problem with OPTN policy is that it’s vague. It’s policy, but it doesn’t make anything black and white, it doesn’t truly give anybody anything to stand on when it comes to asking for enforcement of things.” As a smaller agency, HRSA historically did not seem to have the staffing capacity to deal with complaints. If someone complained to them, they turned it over to UNOS, illustrating the circularity of OPO oversight, as well as the regulatory capture, which has created the facade of oversight without any actual enforcement.
While very few people we spoke with knew about the CMS complaint process, a former CMS worker confirmed that there is a system for CMS to log complaints called the Automated Survey Processing Environment (ASPEN).129 CMS gives ASPEN access to select management and surveyors (Regional Office and State Agency) for use in managing complaints across all facilities including OPOs and Transplant Centers. When a complaint gets entered into ASPEN, Quality and Safety Oversight Group (QSOG) reviews and decides when and if to visit the site. QSOG then goes through steps to determine if the site is within compliance, and if not, it works with the site to get it back in compliance. (See CMS Complaint Map.) If the site fails to correct itself, CMS can ultimately create a public notice of termination, though that has never happened in nearly 40 years. Neither the public nor Congress has access to ASPEN, making it impossible to audit complaints filed and the manner in which CMS responded.
CMS Complaint Map
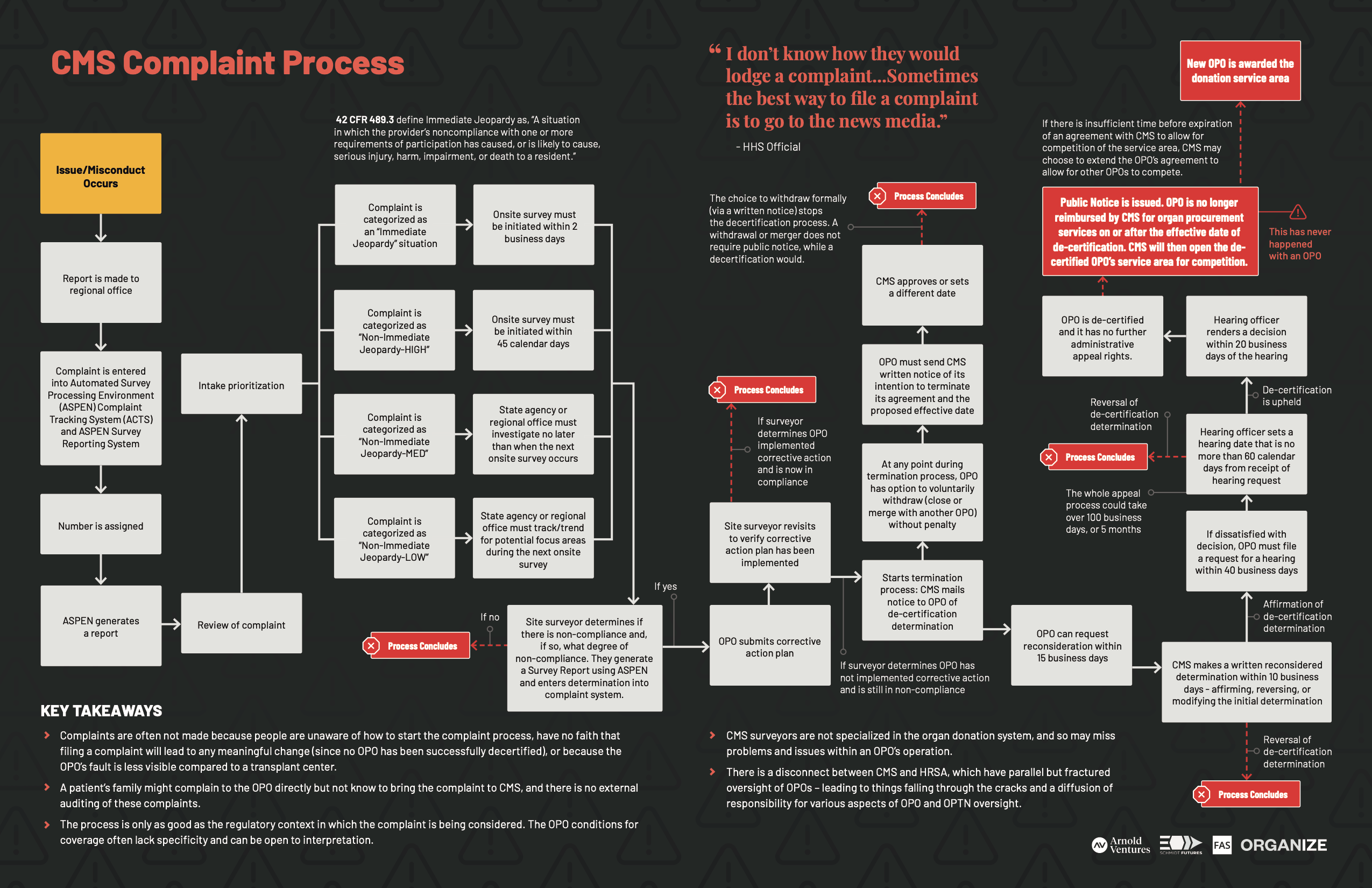
Download the “CMS Complaint Process Map” PDF
CMS intercedes when UNOS places an OPO on probation and/or finds them “not in good standing,” the latter of which, again, has only happened two times. In these instances, CMS cannot take action based solely on what UNOS finds; rather, CCSQ conducts a separate investigation into whether the OPO violated CMS regulations. According to an HHS official, CCSQ does not look at whether an OPO is operating efficiently or financially appropriately.130 The compliance aspects – from the CMS side versus the OPTN side – are largely divorced from each other. Most people we spoke with, both inside and outside of government, expressed little faith that filing a complaint would lead to meaningful change. In fact, no OPO has lost its government contract in the nearly 40 years the system has operated, including:
- the Miami OPO, Life Alliance Organ Recovery Agency, which failed to report a fungus in a uterus before sending it off for transplant, nearly killing the recipient;131 had a supervisor allegedly physically assault a nurse and doctor for speaking out against insufficient staffing and training;132 and was flagged in a peer review process for issues in management, staff training and collaboration with regional transplant hospitals133
- the Alabama Organ Center, whose leaders went to federal prison for receiving $500,000 in kickbacks from a funeral home134
- Kentucky Organ Donor Affiliates, which recovers only 1 in 4 potentially viable organs135
- the Los Angeles OPO, OneLegacy, which inappropriately spent taxpayer money on things like football tickets and parade expenses,136 was scrutinized by press for unethical tissue recovery practices137 and then tried to obstruct the Los Angeles Times investigation138
- the South Carolina OPO, We are Sharing Hope SC, which killed a lung recipient and injured a liver recipient by incorrectly identifying blood type and was later found to have problems with their blood-typing policies139
- Donor Network of Arizona, which took someone’s corneas without consent140
- Nevada Donor Network, which misled a donor family and “took more tissue than what was authorized” according to a lawsuit141
Two of these OPOs received a designation of “member not in good standing” by UNOS (Life Alliance Organ Recovery Agency and Nevada Donor Network) but the others have received no such designation or repercussions. Given UNOS and AOPO’s continued assertion that the success of the organ donation system relies on “maintaining public trust,” it is vitally important that the public have faith that OPO improprieties are identified and remediated.142
The ineffective complaint system and fear of retaliation results in a stagnant organ donation field that is more concerned with self-preservation and maintaining the status quo than improvement and innovation. This means that the 110,000 patients on the waitlist who desperately need more organs to be recovered will continue to suffer, and too many people, disproportionately patients of color, will die without the transplant they need.143
Transparency Around Conflicts of Interest
Nearly every person we spoke with alluded to conflicts of interest within the current organ transplant system. “The entire UNOS is a conflict of interest,” expressed one government official. Many of the UNOS board members are OPO leaders and transplant surgeons – representing the institutions for which they are making policies. Furthermore, many of the OPO leaders on the UNOS board come from underperforming OPOs, so there’s a disincentive to create stricter performance reviews. Of the nine OPOs represented on the OPTN/UNOS board,144 three are failing the new performance standards, including Iowa Donor Services which is notably among the worst performers in the country.145 Similarly, out of the several members on the MPSC who are directly connected to an OPO, nearly half come from an OPO that is failing the new performance standards.146
“The entire UNOS is a conflict of interest”
- Senior Government Official
OPO members on the MPSC were the ones who created UNOS’ OPO performance review process.147 Since underperforming OPO leaders are the ones creating their own performance review process, it’s essentially the “fox guard[ing] the hen house” as Senators Chuck Grassley and Todd Young wrote to the HHS Inspector General in 2019.148 Currently, the UNOS board has the same members as the OPTN board. This violates explicit direction from the Government Accountability Office (GAO), which in 2018 wrote that UNOS must maintain a separate board from the OPTN. The office explained its rationale, writing, “HRSA staff regularly observed instances where it was not clear whether the OPTN Board was acting on an OPTN-related matter or a contractor-related matter. The one-to-one correspondence between these boards amplified the lack of clarity.”149 150
Several people we spoke with explained that because it’s a small community, it can be hard to find subject matter experts to fill leadership positions without any existing conflicts. But, currently, the system requires an insufficient level of transparency around such conflicts. Thus, there is no pressure for individuals with conflicts of interest to make those interests transparent, or for the government or the public to evaluate the extent to which such policymaking processes are compromised by competing loyalties.151 According to one OPO CEO, “when OPTN Board members do disclose their conflicts of interest, it’s very informal. They talk about taking off their ‘transplant center hat’ and putting on their ‘OPTN hat.’ They know their interests compete, but they simply think they are entitled to the conflict.” In fact, as recently as September 2020, a Federal Judge ruled that emails between “UNOS decision makers and affiliates [showed] clear preferences for policy outcomes which the Court previously characterized as ‘arguable evidence of bias, or at least, individuals’ sporadic expressions of bad faith or agenda.”152 In UNOS’ response to the Senate Finance Committee oversight letter, they list the ways in which conflicts of interest are disclosed for the OPTN board and MPSC, yet it is questionable how effectively implemented these policies are, given we understand all disclosures are self-reported, and that the definition of such conflicts is often overly narrow.
Another version of opacity around conflicts of interests is the history of public comments made on the proposed rule and other Hill lobbying that were purportedly from unbiased third party medical professionals, but who were actually paid advisors to various OPOs. For example, the Connecticut and Vermont Medical Examiners provided comments arguing against death certificate accuracy for use in the proposed rule,153 an assertion upon which AOPO rested much of its opposition to proposed reforms. However, a Los Angeles Times investigation outlined how OPOs have co-opted medical examiners for lobbying purposes through direct payments and other gifts and event sponsorships.154
Similarly, AOPO cites “Emory Transplant Center” in their document against the proposed rule.155 However, this comment was actually written by an individual doctor who works at Emory and was paid $100,000 that same year as a board member of LifeLink Foundation,156 which operates three OPOs that were all considered failing in the notice of proposed rulemaking (NPRM). This financial conflict of interest calls into question the motivation behind the doctor’s argument that closing failing OPOs would be too disruptive, given that, at the time this doctor submitted their comment, the OPOs compensating them appeared to be at risk of being decertified.157 LifeLink has subsequently come under Congressional investigation for various potential abuses, including its anti-patient lobbying tactics.158 This underscores the need for HHS and Congress to have more readily accessible visibility into the financial connections across the various transplant stakeholders that engage in lobbying and advocacy, including financial ties to OPOs or their partners, as well as the need for the ongoing Congressional investigation into LifeLink Foundation to assess the extent to which LifeLink employed such tactics in lobbying against HHS’ OPO reforms.
Finally, another theme we heard repeatedly was that individual OPO leaders are able to concurrently serve in multiple conflicting roles across the organ donation system, effectively consolidating power rather than checking it. For example, consider Alexandra Glazier, CEO and Board member of the New England OPO, which was highlighted in investigative reporting by the Project on Government Oversight (POGO) as one of the most aggressive OPOs in lobbying against HHS reforms for OPO accountability.159 Despite the OPO being the subject of an oversight investigation from the House Committee on Oversight and reform into potential conflicts of interest and anti-patient lobbying,160 Glazier serves in the following positions:
- Councilor on the OPTN/UNOS Board of Directors
- Chair of the UNOS Policy Oversight Committee
- a member (and past Chair) of the AOPO Legislative and Regulatory Affairs Committee
- a founding member of OPO lobbying group Organ Donation Advocacy Group
- a Board member of Donate Life America
- Member of the SRTR Review Committee
- UNOS MPSC, formerly
- UNOS Ethics Committee, former Chair
- Member of the UNOS Geography Committee161
- HHS’s Advisory Committee on Organ Transplantation (ACOT), previously
Even though this person is openly and actively lobbying against new accountability measures, she’s in a position where she can influence the very institutions that are supposed to keep OPOs accountable.162 Even though her influence is suspect, she’s serving positions where she’s supposed to:
- “lead the strategic coordination of national policy development” for OPTN/UNOS163
- set the lobbying agenda for two separate OPO industry lobbying groups
- advise the SRTR on how to risk-adjust data CMS relies upon to evaluate OPO performance
- influence national messaging around organ donation via Donate Life America.
Previously she advised HHS on OPO policy and until recently was involved in settling complaints about OPO misconduct for UNOS.
Additionally, senior staffers and board members at Glazier’s OPO serve as:
- Chairperson for the American Association of Tissue Banks (AATB) Board of Governors164
- Member of the the AATB Quality Council
- Chair of the Donate Life America Data and Research Committee
- Member of UNOS OPO Committee
- UNOS President, previously165
- Advisor to a current National Academy of Medicine study to, in part, “Consider the development of a new, standardized, objective, and verifiable donation metric to permit the transplant community to evaluate DSAs and OPOs and establish best practice.”166 While these various organizations can provide the veneer of independent validation, in effect there is often just a small group of individuals coordinating all efforts. According to an OPO employee we talked to, “One week we all meet at a conference for AOPO; the next week we meet at a conference for UNOS; and the next week we might be on some kind of OPO advisory committee for the American Society of Transplant Surgeons. But it’s all the same people, all we change is the letterhead for our statements.”
Communication Across a Diffuse Governance Structure
A common theme we heard throughout our research was that there’s a disconnect between the various structures that each have an arm in organ transplantation oversight. According to one government official we spoke with, groups within CMS do not routinely talk, collaborate, nor readily share information with each other. Each has a separate function and seems to only focus on that specific function. Within CMS, there are three main groups involved in OPO oversight:
- Clinical Standards Group (CSG) writes conditions for coverage
- Quality and Safety Oversight Group (QSOG) enforces conditions/implements policies
- Survey and Operations Group (SOG) is out in the field doing surveys and certifications
“There is a disconnect between the team that writes the rules and the team who actually enforces it [within CMS],” said one HHS official. In 2019 a “longtime Department of Health and Human Services official” told the Washington Post that CMS’ inability to hold a failing OPO accountability to the current regulations is because CMS’ oversight is “based on a metric that is not effective. It’s just another indication that [CMS is] unclear on what they are doing when it comes to providing oversight to the OPOs and it’s continuing to provide confusion in the field.”167 CMS did have periodic meetings with HRSA, but they were not frequent. HRSA was more focused on the OPTN, while CMS is focused on OPOs. They don’t seem to be intersecting on the piece where the OPTN is supposed to be overseeing OPOs.
The lack of communication and collaboration means that often oversight is not carried out effectively. And agencies often miss patterns that could be identified and addressed through a more comprehensive and collaborative analysis. For example, one department looking at OPO outcome metrics may not make the connection that poor OPO outcomes may come from inefficient resource allocation of staff, which would be evaluated by the financial person in another department. “There needs to be cross pollination between the different departments overseeing the OPOs and OPTN,” said one government official. “Right now the problem is that there’s no one in charge of everything.”
“There needs to be cross pollination between the different departments overseeing the OPOs and OPTN. Right now the problem is that there’s no one in charge of everything.”
- government official
UNOS typically does not share data with OPTN members, and OPOs do not share data with each other. This results in an opacity around how each OPO is actually performing compared to its peers and prevents OPOs from readily learning about what poor practices to avoid or best practices to adopt.168 “There’s no alignment across the industry to get more transplants. There’s an alignment for self-preservation,” said one OPO leader.
Financial Incentives
As UNOS noted in their response to the Senate Finance Committee letter, it does not conduct financial audits of OPOs.169 Given that CMS also does not regularly audit OPO finances, there is a large gap in oversight of proper resource allocation within OPOs.170 This further underscores the need for HHS to reconsider OPO reimbursement structures; OPOs are the only major program in all of health care that is fully reimbursed for all costs, even those not directly related to organ recovery.171 As there are currently no meaningful mechanisms to audit OPO finances, it is all the more troublesome that OPOs are still allowed to operate on a reimbursement structure which historically has been susceptible to fraud, waste, and abuse.172
The current OPO compensation model does little to incentivize OPOs to procure more organs. CMS reimburses OPOs based on self-reported costs, and not based on any kind of performance metrics. The SAC fee, which each OPO calculates individually with no transparency on what is used for its calculation, has no government oversight, and continues to rise dramatically each year. SAC fees vary by as much as 100% across OPOs for the same organs.173 One OPO leader shared with us that their cost reports to CMS had never been audited in at least 12 years.
OPO Financial Compensation Model
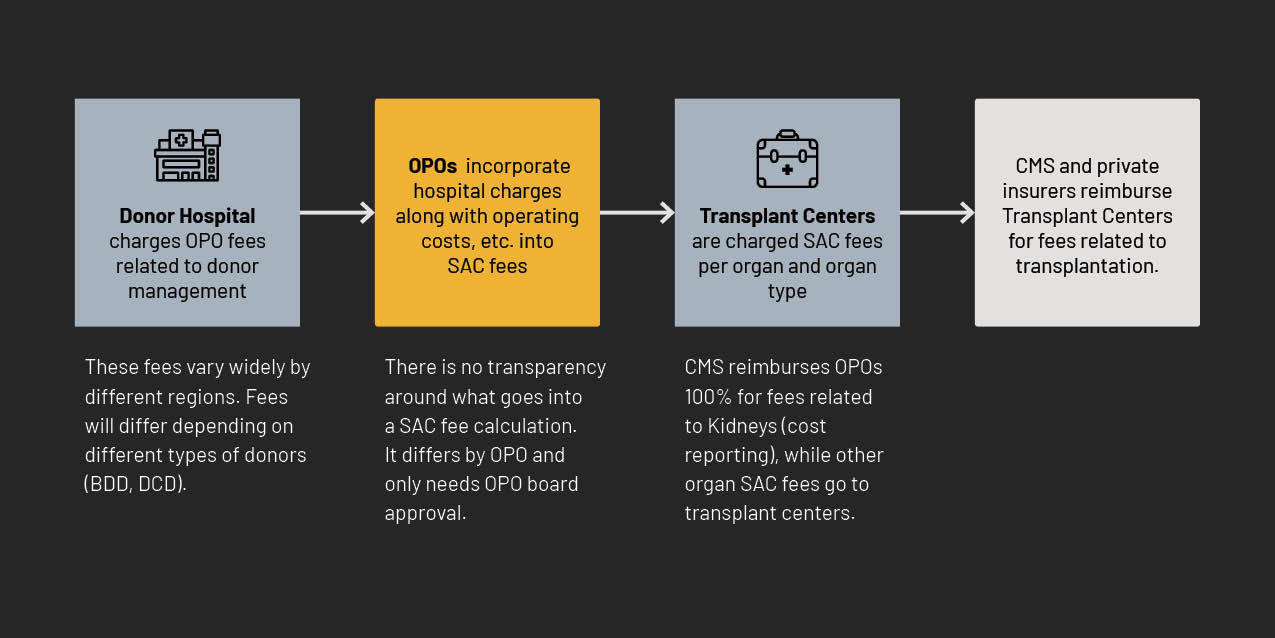
Download the “OPO Financial Compensation Model” PDF
Between 1996 and 2014, the cost to Medicare for acquiring organs increased by 253 percent, according to an analysis.174 However, in that same time frame, the volume of donors increased by only 57 percent, and the volume of transplants increased by only 45 percent. Throughout the country, total organ acquisition costs are about $3 billion annually.175
This 100% reimbursement structure has also allowed OPOs functionally unlimited resources with which to fight against reforms.176 For instance, investigative reporting from the Project on Government Oversight (POGO) found OPOs and UNOS “ratcheted up [their lobbying expenses] dramatically in apparent response to [proposed OPO reforms].”177 In a recording obtained by POGO, one OPO executive, presenting at an official UNOS conference, stated that OPOs were “fortunate” for COVID, which afforded OPOs more time to hire lobbyists and to organize “a real congressional action campaign” against the proposed rule changes.178 AOPO, the OPO trade association, recently told its members that it planned to change its status from a 501(c)(3) to a 501(c)(6)179 to allow for freer spending on lobbying.180 One former government official we spoke with said, “these 58 organizations are extremely well organized. They are very loyal to each other. They are going to dig their heels in at any change that you want to make.” A former Chief Technology Officer at HHS wrote that “[OPOs are] some of the most obstructionist stakeholders I’ve ever come across.”181
There are also several documented cases of OPOs settling lawsuits out of court, for various complaints ranging from wrongful death, to racial, sex, and job discrimination.182 We’ve heard from a senior government official that since 100% of their expenses are reimbursed, OPOs are able to use taxpayer dollars to settle these lawsuits out of court. This presumably undercuts pressure to not engage in such behaviors, as well as ensures that the government and general public do not learn about problematic practices. CMS should confirm and further investigate whether taxpayer dollars are being used in this way, and as needed, reform policy to disallow OPOs from including such litigation and settlements into their taxpayer-funded reimbursements.
Further, not only do OPOs often ineffectively allocate resources, but there is reason to believe that at least some OPOs may even have other competing priorities.183 In addition to solid organs, OPOs also recover human tissue, and within the last 15 years, procuring tissue has become much more financially lucrative, and thus popular, for OPOs. According to the Los Angeles Times, “80% of [the OPOs] were harvesting tissues and sending them in exchange for fees to tissue processors and distributors, many of which are for-profit companies. Today the selling of human tissue is a multibillion-dollar global business.”184 While the FDA provides clinical regulations on tissue procurement, it does not oversee the financial and resource allocation aspects of tissue, nor does anyone evaluate potential conflicts of interest. As one former OPO leader we talked to described, “I’ve had bosses tell me that our OPO is ‘tissue-first, organ-second. Tissue crowds out the mission [of procuring organs].” Since tissue is so financially lucrative, OPOs often have an incentive to direct more of their resources and staff on tissue procurement at the expense of organ procurement. Additionally, since OPO executives and board members don’t have to disclose personal financial interests in entities the OPO partners with, a CEO may have an (undisclosed) personal incentives to prioritize tissue over organs, even if it’s not even in the OPO’s financial or mission-driven interests.185
UNOS also charges a double fee to transplant centers for every patient added to a waitlist. In addition to an “OPTN Regulation Fee,” which is part of a mandate approved by HRSA, UNOS has also simply asserted its right to charge a parallel “UNOS Registration Fee,” which is not part of the mandate. UNOS earns about $58 million a year, and as the New York Times Editorial Board explains, “because most of that money comes from patient fees, there is more of an incentive to add patients to the waitlist than to secure organs for them.”186
Technology and Security
Most people within the organ donation system rely on the UNet℠ platform – one of the key tech systems within the community. The UNet℠ system is built and managed by the current OPTN contractor, UNOS, and is closed and proprietary. A leading healthcare technology executive described UNOS’ technology inner workings as “literally duct tape.”187 Older systems with unstructured and difficult-to-maintain source code increases the surface area for security vulnerabilities. Additionally, the proprietary nature blocks any chance of innovation or competition from knowledgeable outsiders.
Even though UNOS was paid to build and implement their system with government and taxpayer money, UNOS refuses to allow government access to servers, code, and much of the data powering the platform. According to people we spoke with in the community, they quoted a price of approximately $20 to 60 million to sell the code back to the government – even though it was initially funded by the government.188 Requiring the government to buy back code for millions even though the government paid for the system’s development helps UNOS hold their position. It also hurts OPOs, and ultimately patients, who could benefit from better innovation and competition that open-source platforms can facilitate.189
HRSA, the agency formerly tasked with overseeing the OPTN contractor (soon to be OASH), had few technical staff who could effectively assess UNOS’ technical capabilities and practices. “We don’t have the in-depth IT staff to have an understanding [of] whether the things [that] are being built are good,” said one HHS official.190 Additionally, UNOS’ backend data is stored in physical data centers that are maintained by UNOS. This poses security risks in the case that they are damaged by external factors. Most modern government software, as well as private industry, leverage commercially available cloud providers, many of which are certified to run government maintained workloads.
Several interviewees we spoke with mentioned the system downtimes, development inefficiencies, and implementation delays within UNOS technology. For example, in 2013, a new policy update allowed organs from deceased HIV+ donors to be recovered and transplanted in compatible HIV+ recipients. But it took years, according to interviewees, for this change to be reflected in UNOS’ match run program, DonorNet. This made it incredibly difficult for patients to actually benefit from the new policy.
“Every coordinator’s ability to do their work is impacted by [DonorNet] not being a better tool.”
- OPO leader
Recommendations
Based on our findings about the gaps and conflicts within the oversight of OPOs and the OPTN, we share below a series of recommendations for how these issues may be addressed. There is a clear opportunity for increased Congressional oversight into OPOs and UNOS, with the opportunity to more fully understand – and then subsequently address – the structural problems plaguing the organ donation industry.
Themes in recommended additional oversight include:
- UNOS Oversight: including auditing of UNOS MPSC records for conflicts of interest and OPO protectionism.
- OPO Finances: including misspending of taxpayer resources; OPO financial conflicts of interest and self-dealing, especially as it pertains to tissue recovery; failures of the current 100% reimbursement model for OPO to properly incentivize organ recovery.
- OPO Lobbying: including exploitation of loopholes to reimburse OPOs for lobbying activities, such as via board compensation for politically connected board members.
- Racial Equity: including differential treatment of – and outcomes for – patients and donor families of color.
- UNOS technology: including auditing of security vulnerabilities in the current system and more transparency into the code behind the different technology platforms.
Many of our recommendations are geared towards a future “Office of Organ Policy,” as called for by patient advocates, that can implement and produce outputs from identified issues. But we need all parts of Congressional oversight to increase the scrutiny on OPOs and the OPTN to identify those issues and provide inputs for what the Office of Organ Policy should work to address.
Build an Empowered Office of Organ Policy
When asked what is one thing that would be crucial for improving oversight and governance in this system, one industry expert said: “A more unified, visible, and substantial entity within HHS to oversee the system. It would be empowered to establish the goals and objectives of the system and be empowered to collaborate with governmental and non-governmental entities to get the best resources and abilities available to get them to bear on issues of allocating scarce resources and allowing more organs to be available.”
We believe a new Office of Organ Policy (OOP) can be this unified and empowered entity for the organ donation and transplant system.191 To be empowered to take on the role of consolidating and effectively improving oversight of OPOs and the OPTN, OOP will need to have the authority to enact change, the right staff, skill set, and a roadmap for which issues to prioritize.
If the Office of Organ Policy were based in the Office of the Secretary or in Office of the Assistant Secretary for Health (OASH), the authority to enact change can come from the OPTN Final Rule, which gives the HHS Secretary (and its designees) broad leeway to gather the information necessary to carry out their responsibilities.192 Additionally, HHS lawyers could interpret NOTA to focus on its goals and objectives, rather than the OPTN structure.
The OOP needs to be staffed with not only the right skills and knowledge for the wide variety of functions needed to oversee the organ donation field, but those experts also need to be free of any vested interests with UNOS or OPOs in order to avoid further entrenchment of conflicts of interest. As one OPO CEO described, “[HHS] would have more confidence if they had experts around them who didn’t have a vested interest in winning the contract. They need to be able to scan the horizon, identify opportunities to improve governance, and deploy the improvements in a systematic way.”
Skill sets the OOP should prioritize in engaging talent include:
- Technologists to interface with the OPTN organ matching technology (currently DonorNet) and continually monitor it for efficacy and improvement as well as system and technology accountability;
- Clinical experts in organ transplantation to oversee clinical standards, policy, and allocation algorithms;
- Financial experts, including with expertise in forensic accounting, to evaluate OPO and OPTN resource allocation, and flag inappropriate reimbursement claims as needed;
- Health care financing experts to evaluate alternative OPO financing models with the goal of aligning OPO financial incentives with the goals of patients and taxpayers (i.e., maximizing life-saving organ recovery and transplantation);
- Leaders who can help address racial inequities in the organ transplant system
- Merger and Acquisition expertise for OPO consolidations, since that is the most likely way to decertify an OPO moving forward;
- Systems thinkers who will focus, urgently and holistically, on addressing systemic rather than acute problems;
- Analysts with the ability to evaluate policies and regulations – HRSA over-relied on external contractors like the SRTR, especially given potential SRTR conflicts;
- Market design economists to optimize living kidney donor chain systems;
- Open data experts who can coordinate with external researchers.
The OOP may be able to do things that HRSA previously could not via authority as a designee of the HHS Secretary. We suggest leveraging this opportunity by prioritizing hiring experts with the skill sets outlined above to tackle these longstanding problems. For example, if the OOP can get access to audit UNOS’ tech stack code (which HRSA previously could not), it makes sense for OOP to hire digital services experts; similarly, if the OOP can access UNOS MPSC files, it makes sense to engage oversight experts.
The OOP should provide a cohesive strategic vision and ensure alignment of practices and policies across all of HHS consistent with unified goals. In terms of the staffing structure, the biggest difference between the OOP and the current diffuse governance structure will be that there is a director of the OOP who oversees all aspects of organ donation and transplantation. The ultimate responsibility would lie with this role to ensure that nothing is being missed and that the system is continually improving. In the current system, that responsibility does not lie with any single role. As a result, different entities only carry out the minimum of what they are tasked with, rather than taking a holistic view and proactive stance to address systemic issues.
Under the OOP, there should be different staff overseeing transplant centers, OPOs, and the OPTN. Transplant centers and OPOs should have different staff because each requires different types of knowledge. Staff overseeing the OPTN and OPOs should be different to maintain separation of undue influence of the OPTN contractor on government OPO policies. Above all, the OOP should have a strong conflict of interest clause that says anyone in a leadership position at the OOP cannot then go on to work at or lobby for an OPO or the OPTN contractor afterward.
Once the correct staff and structure are in place, there are several key steps that the OOP can take in their first few months to improve oversight and efficiency in the organ transplant system. Many of these actions will require the OOP to coordinate with various agencies involved with the OPTN ecosystem. The OOP should become the conductor and coordinator of all agencies as it relates to OPTN and OPO issues. From the onset, the OOP should strive for transparency in their work and plan to give frequent updates and reports to Congress on the actions listed below.
- Tech Audit: Utilizing tech experts, OOP should do a thorough audit of the UNOS technology stack and pursue access to the code on which the technology is built. This code should be held up to modern technology standards and examined for security vulnerabilities. The OPTN contract should also be reviewed and the next RFP should be built to open up competition for potential new vendors.193
- Complaint Audit: OOP staff should request and audit the complaint history handled by CMS and MPSC to evaluate what actions were taken or not taken for every complaint and re-open cases as needed. This should include investigating any conflict of interest194 for each MPSC member at the time when each complaint was processed. The goal is to take action on what may have been previously missed, as well as identify trends among the reports for systemic issues across OPOs.
- Revise Complaint Process: OOP staff should initiate a user-centered design project to both rebuild the complaint system and provide public education to all stakeholders (patients, transplant centers, hospital staff) on how to access and report complaints.
- Financial Audit: OOP staff should work with governmental partners to do regular audits of each OPO’s finances to identify if there is ineffective resource allocation (such as understaffing of organ procurement, especially as compared to staffing for tissue procurement) or improper reimbursement claims. This should include reviewing executive compensation and how that compares to the OPO’s performance, and advise on alternative reimbursement models (including for executive compensation) that would better harmonize resource allocation incentives with the goal of maximizing organ transplantation.
- Public Website: Given each OPO’s regional monopoly status, the OOP should aim to bring more transparency to OPO performance and how that affects patients. One way to do this is to create an easily accessible public website showing OPO performance data and compliance according to the recently finalized outcome measure changes and its implications for patients.
- OPTN Policy: Review all current OPTN policies to evaluate if and how they contribute to racial inequities, including considering transparency requirements for how OPOs respond to potential donors. Review conflict of interest policies and how they’ve been implemented for both the OPTN and OPOs. For conflicts of interests that are unavoidable, there should be full public transparency made for every instance.
- Data Access Policy: Coordinate with SRTR and establish data access policies that would enable researchers safe access to OPO and OPTN data without the need for OPO or OPTN contractor approval. This is meant to encourage and enable independent academic analysis of OPO and OPTN performance, to bring fuller transparency and help continually improve the system.
- Strategies to Optimize Organ Donation: OOP staff should further engage data experts to develop more effective and standardized strategies for optimizing organ donation. This should include identifying opportunities for better integration between donor hospital and OPO data systems to support faster identification of potential donors, and work with CMS to update OPO Conditions for Coverage relating to OPO processes. And it should include revisiting and updating CMS’ definition of “Urgent Need” for use in decertifying failing OPOs.195 Along with OPTN oversight functions, the OOP should coordinate on several OPO governance functions currently held by CMS. At the top of this list is OPO certification, as specified in the Public Health Service Act. OOP would then set conditions for certification and CMS’ conditions for coverage would point to the new certification conditions. Transferring this function to OOP would enable specialized auditors to focus solely on OPO site surveys and better understand the nuances of the organ donation system so that they can more readily identify problematic practices. Relatedly, the OOP should coordinate with the Center for Medicare on reforming OPO reimbursement policies to better incentivize OPOs to allocate resources properly and get more organs transplanted.196
Another function is for the OOP to oversee quality measures and improvement programs pertaining to organ donation, including all complaints, learning collaboratives, and innovative projects. With the consolidation of these functions into one office, there can also be a consolidation of legal counsel. According to one government official we spoke to, “right now there are various legal counsels who each serve a different agency, and all only know that agency’s part of the fractured system. Someone knows the ‘CMS part.’ Someone else knows the ‘HRSA part.’ What we need is someone who understands the ‘organ donation part.’” A consolidated view including all of the legal components of regulations and laws pertaining to OPOs, transplant centers, and the OPTN, and understand how any policies would impact each individually as well as broader systems dynamics across them would greatly benefit the policymaking process.
Above all, it is important to clearly define the mission and directive of the OOP in order to empower and encourage its staff to do what it takes to constantly improve the organ transplant system to recover and transplant organs, rather than maintaining the status quo.
Decrease Conflicts of Interest on Governing Boards
We strongly believe the organ transplant system could reduce conflicts and instill trust by making the following changes:
- Separate the governing boards for the OPTN and its contractor (currently UNOS)197;
- Prevent UNOS board members from taking HHS regulatory oversight roles for at least 5 years;
- Strengthen currently ineffective regulation around OPO governing boards including their qualifications, representation, practices, and roles.
“We built this system based on how transplantation looked in the early ’80s. It’s not that anymore. We should look at reforming the OPTN to meet what the needs are now.”
-HHS Official
Improve Survey and Complaint Process
Anonymous complaints should be able to be submitted in a simple, accessible, and clear way. HHS should do “root cause analysis”198 of incidents as part of their recertification process and site reviews. And the government should have access to all complaints or even shift its role to become the interface for all incoming complaints. As noted above, the OOP can have a heavy role in this change by initiating this rebuild and by seeding education campaigns for industry stakeholders about how to access and use the complaint system.
Increase Transparency and Data Visibility
According to the Scientific Registry of Transplant Recipients (SRTR)’s latest annual report of 2018 OPO data, 4,994 organs were recovered but discarded in 2018, which is an increase from 4,813 in 2017.199 While discard codes provide some indication of why these organs were not transplanted, it lacks details and transparency into what decisions were made that led to this discard. We can see that the most frequent reason listed for discard is “620: No recipient located - list exhausted” which should be analyzed further to understand how these over 1,500 discards (and resultant shadow deaths) could have been avoided. There are also nearly 800 organs that have a discard code of just “other.” This data is not disaggregated by OPOs in this report, nor shows a comparison of the highest and lowest performing OPOs so that OPOs can truly understand how they are performing compared to peers. SRTR’s OPO-specific reports, which do show data by donation service areas (DSAs), do not list the reasons for discards.200
Reporting data on each organ should include what decisions were made by whom that resulted in an organ not being transplanted. For example, if a transplant center denied an organ because an OPO refused to do a test on the organ that would enable the transplant center to better decide if the organ is viable, that should be documented via a refusal code with detailed comments.201 This data should be available to the public so that trend analyses can be made on whether certain OPOs have done that for multiple organs.
Conclusion
Every time a heart goes unrecovered because an OPO is understaffed or a kidney gets lost due to inadequate transportation processes, someone dies on the organ waitlist. These deaths rarely generate action from oversight bodies, but they are devastating to families and loved ones. There are 110,000 people currently languishing on the organ transplant waitlist, and they deserve an organ donation system that is optimized and held accountable.
We’ve seen promising evidence of OPOs turning around when they feel pressure from oversight bodies. For example, in 2019, Senators Chuck Grassley (R-IA) and Todd Young (R-IN) sent an inquiry to the Inspector General about an OPO in Indiana – specifically asking if Indiana Donor Network charged taxpayers for private jet travel unrelated to organ recovery.202 203 In apparent response to this oversight pressure, the OPO engaged external consultants to improve its organ recovery operations. In the subsequent 12 months, the OPO increased donation rates by 44%; and it increased its ranking from 51st to 18th among the nation’s 57 OPOs,204 underscoring vast underperformance at OPOs. Importantly, the increased donation rates seem to have resulted simply from the OPO exerting more effort; over this period of improvement, the OPO approached 57% more families for donation than it had over the previous 12 months. This suggests that, in the absence of oversight, the OPO seems to have flagrantly ignored its mandate to pursue all possible donation opportunities, and it is entirely likely that hundreds of additional lives have been saved over the past year as the direct result of Senators Grassley and Young simply asking oversight questions.205 This is an encouraging example of how elected officials can best serve their constituents. However, without more and sustained Congressional oversight and regulatory pressure, shadow deaths will remain high, and even OPOs that have improved amidst heightened scrutiny could backslide into previous practices. For this reason, we implore the U.S. government to take steps to hold OPOs and UNOS more accountable and heed these important recommendations.
Appendix
A. Documented Cases of OPO Misconduct
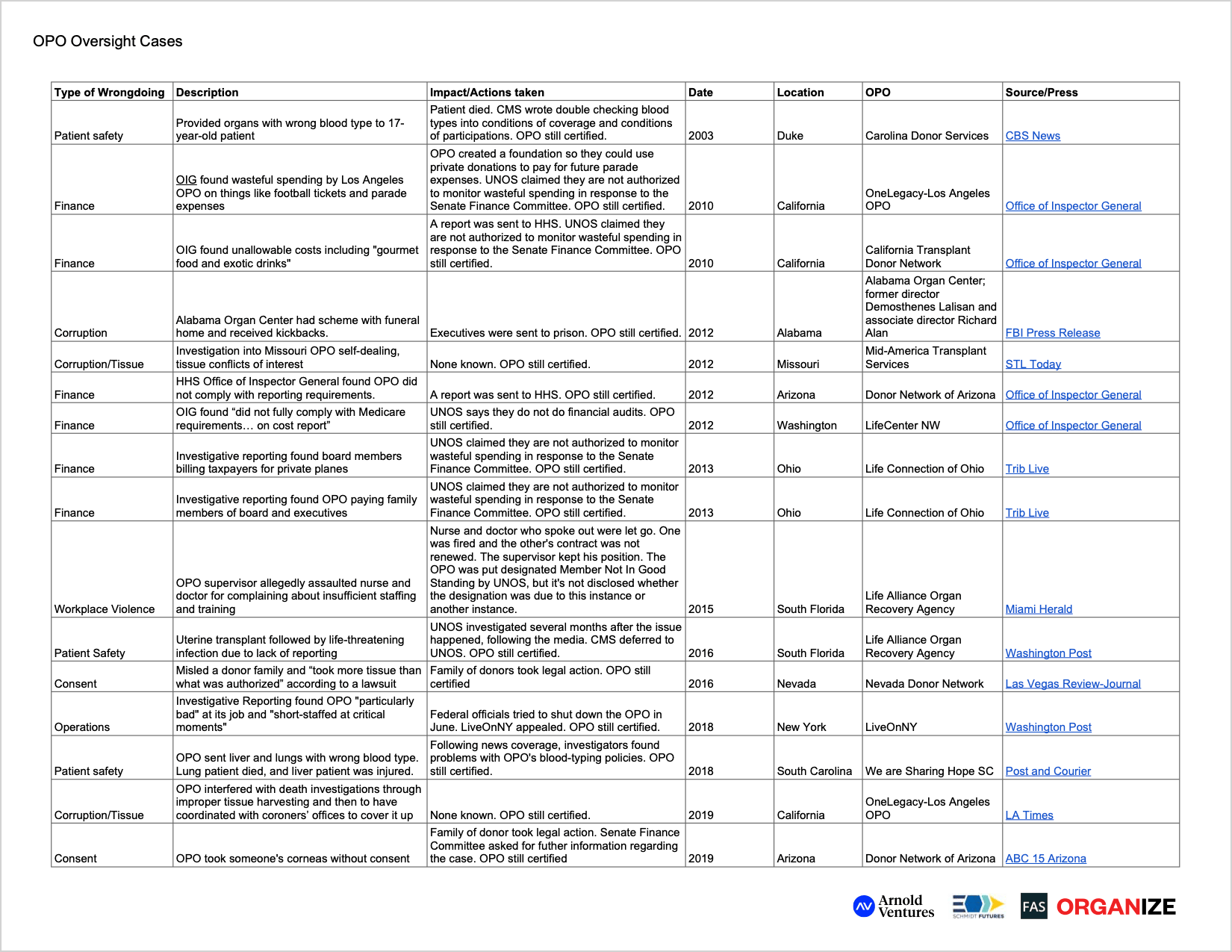 View the Documented Cases of OPO Misconduct PDF
View the Documented Cases of OPO Misconduct PDF
B. UNOS Response to Senate Finance Committee Inquiry Letter
 View the UNOS Response to Senate Finance Committee Inquiry Letter PDF
View the UNOS Response to Senate Finance Committee Inquiry Letter PDF
Additional information on how UNOS changed their website after SFC inquired about their oversight practices:
- UNOS website as of February, 2020, before receiving oversight inquiry from Senate Finance Committee:

Updated UNOS website, as of March 2020, upon responding to the Senate Finance Committee that UNOS is not responsible for the oversight functions about which SFC inquired:
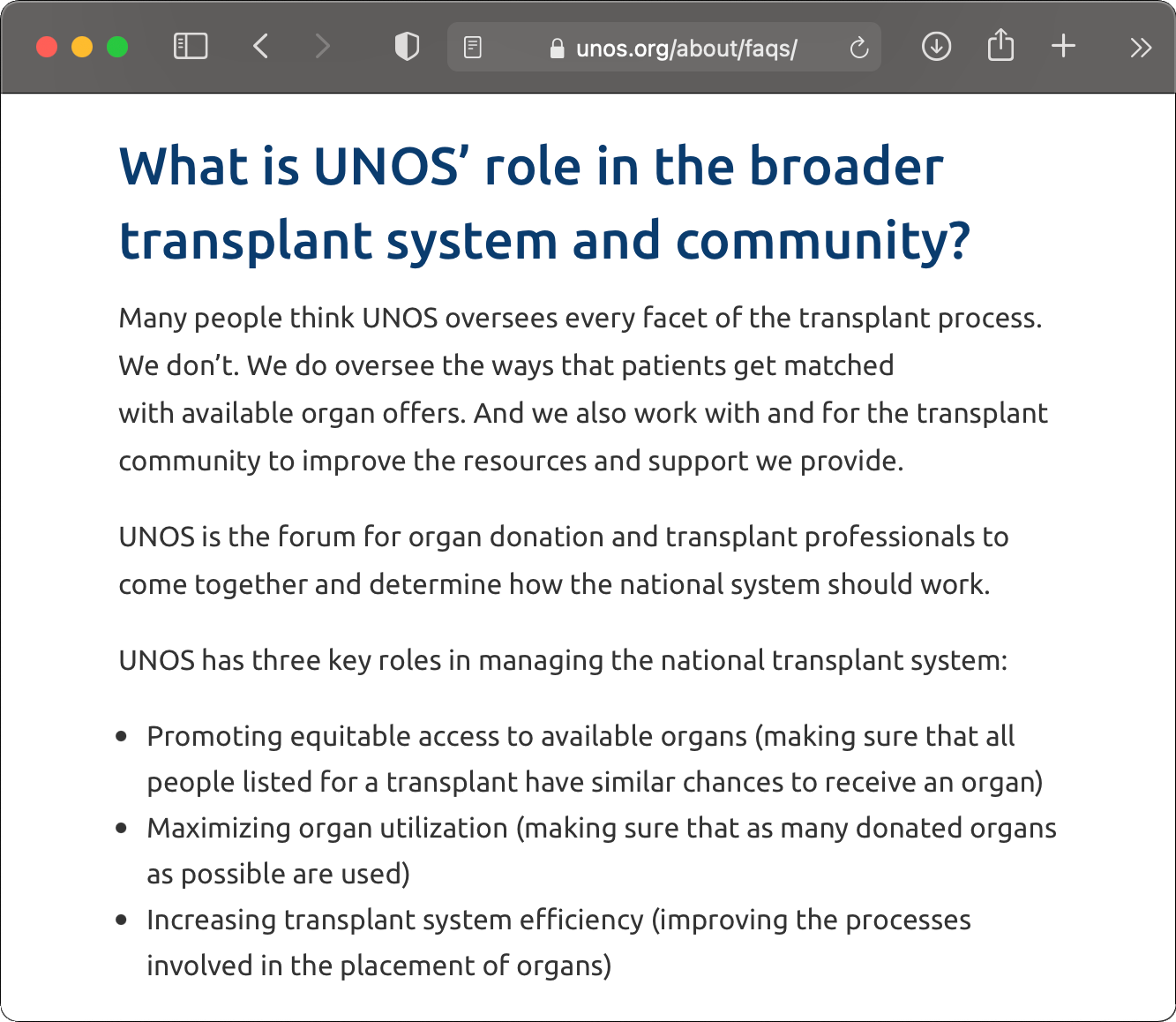
C. Glossary of Terms
 View the Glossary of Terms PDF
View the Glossary of Terms PDF
Notes
Senate Finance Committee Letter to HHS, 2020.
Congressional Letter from Katie Porter and Karen Bass to HHS, 2020.
Congressional Organ Donor Letter to HHS, 2020.
-
“Reforming Organ Donation in America: Saving $25,000 Lives per Year and $13 Billion in Taxpayer Funds over Five Years,” The Bridgespan Group, 2019. ↩
-
“Opinion · Many die waiting for organs. The Trump administration could help.” Washington Post, 2020. ↩
-
As of 1/1/2021, LifeChoice Donor Services and New England Donor Bank have merged, changing the total number of OPOs from 58 to 57. NEDS Successfully Merged OPOs ↩
-
According to data from HHS, OPO performance varies by as much as 470% ↩
-
“Reforming Organ Donation in America: Saving $25,000 Lives per Year and $13 Billion in Taxpayer Funds over Five Years,” The Bridgespan Group, 2019. ↩
-
“Inequity in Organ Donation · The Costly Effects of an Outdated Organ Donation System,” The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
OPO claimed to CMS that they recovered 69% of potential donors in their self-reported data. Congressional Letter from Katie Porter to HHS, 2019. ↩
-
“Reforming Organ Donation in America: Saving $25,000 Lives per Year and $13 Billion in Taxpayer Funds over Five Years,” The Bridgespan Group, 2019. ↩
-
“Heartless: Organ Donation Contractors Lobby Against a Popular Health Care Initiative While Pocketing Pandemic Relief Loans,” Project on Government Oversight, 2020. ↩
-
OneLegacy 990, 2017. Retrieved at CauseIQ; Cited in Congressional Letter from Katie Porter and Karen Bass to HHS, 2020. ↩
-
“Review of OneLegacy’s Reported Fiscal Year 2006 Organ Acquisition Overhead Costs and Administrative and General Costs,” HHS, Office of Inspector General, 2010. ↩
-
“Taxpayers help pay for organ donor groups’ parties, Rose Parade expenses,” Trib Live, 2013. ↩
-
“Following the audit, rather than take steps to boost its clinical performance or strengthen its financial mismanagement and controls, OneLegacy established a foundation in order to use private donations to pay for the majority of the costs related to the Rose Bowl.” From Congressional Letter from Katie Porter to HHS, 2019. ↩
-
“In the rush to harvest body parts, death investigations have been upended,” LA Times, 2019. ↩
-
“Executives coached coroners on how to keep body parts harvesting records secret,” LA Times, 2019. ↩
-
“Heartless: Organ Donation Contractors Lobby Against a Popular Health Care Initiative While Pocketing Pandemic Relief Loans,” Project on Government Oversight, 2020. ↩
-
“Alabama Organ Center kickbacks detailed,” AL.com, 2012. ↩
-
Former Alabama Organ Center Associate Director Sentenced for Fraud, Federal Bureau of Investigations, 2012. ↩
-
“Doctor, nurse: UM administrator assaulted us,” Miami Herald, 2015. ↩
-
“Doctor, nurse: UM administrator assaulted us,” Miami Herald, 2015. ↩
-
“Why did the first US uterine transplant fail?” Washington Post, 2019. ↩
-
“He died when he got the wrong lungs. It wasn’t the only organ error in SC that day.” Post and Courier, 2020. ↩
-
He died when he got the wrong lungs. It wasn’t the only organ error in SC that day.” Post and Courier, 2020. ↩
-
“Organ Procurement Organization (OPO) Conditions for Coverage Final Rule: Revisions to Outcome Measures for OPOs,” CMS, 2020. ↩
-
The November 2020 Final Rule stated that failing OPOs would not be decertified until 2026, but many advocates and elected officials have called on HHS to accelerate this timeline. As U.S. Representatives Katie Porter and Karen Bass noted in their letter to HHS, “We cannot consign 20,000 or more patients to die waiting for organ transplants while federal contractors are not held accountable, and therefore urge you to use the new standards in the next recertification cycle.” From “Congressional Letter from Katie Porter and Karen Bass to HHS,” Congress of the United States, 2020. In December 2020 the White House Office of Information and Regulatory Affairs published its 2021 rulemaking agenda, including “Amendments to Rules Governing Organ Procurement Organizations,” which would provide the opportunity to address the decertification timeline. ↩
-
Based on 2018 data, 12 OPOs are classed as “Tier 2,” which opens them to competition from other OPOs, and 22 OPOs are classed as “Tier 3,” which opens them to decertification. From “OPO Final Rule,” CMS, 2020. ↩
-
“Medicare and Medicaid Programs; Organ Procurement Organizations Conditions for Coverage: Revisions to the Outcome Measure Requirements for Organ Procurement Organization,” CMS NPRM, 2019. ↩
-
“Coronavirus destroys lungs. But doctors are finding its damage in kidneys, hearts, and elsewhere.” Washington Post, 2020. ↩
-
“Dialysis patients are at high risk during COVID-19 outbreak,” Washington Post, 2020. ↩
-
Note: In September 2019 CMS issued a Final Rule to remove outcome measures for transplant centers, which we will continue to monitor, including whether and how this impacts transplant center oversight. ↩
-
In 2019, UNOS made changes to the way a patient’s MELD score is calculated. “MELD exception scores during NLRB transition,” OPTN. ↩
-
“She beat cancer. Now she’s in another fight for her life,” New York Times, 2019. ↩
-
“She Beat Cancer. Now She’s in Another Fight for her Life,” NY Times, 2019. ↩
-
For example, in 2003, there was a very highly publicized case at Duke that ended with a young girl, Jesica Santillan, dying from a blood typing error after a transplant. If the OPTN instituted an automated organ offer process when the technology first became available, this error could have been prevented. It wasn’t until the event occurred in 2003 and HRSA wrote the automated organ offer system into the 2005 contract that DonorNet – the automated matching system – finally came into existence. Similarly, it was only after a case became publicized of rabies getting transmitted through a transplant that HRSA started requiring the OPTN/UNOS to collect data around disease transmission cases. Data collection of specimen and organ labeling errors led to the OPTN creating TransNet, which is UNOS’ software for organ packaging and labeling. As one OPO leader told us, “The community functioned for years completely unaware there was a problem waiting to happen.” ↩
-
“20% of U.S. Transplant Centers Are Found to Be Substandard,” LA Times, 2006. ↩
-
“US transplant overseer sets guidelines,” LA Times, 2007. ↩
-
“Hospital Is Rebuked in Organ Case,” LA Times, 2006. ↩
-
“Transplant Center Regulations—A Mixed Blessing? An ASTS Council Viewpoint,” American Journal of Transplantation, 2008. ↩
-
“OPO Best Practices · The Costly Effects of an Outdated Organ Donation System,” The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
A Congressional notification released stated, “HHS also moves the Division of Transplantation from the Healthcare Systems Bureau in HRSA to OASH, and abolishes the Healthcare Systems Bureau. The reorganization is effective today, Friday, January 15, 2021. We anticipate that most activities will transition within 4 to 6 weeks, but others, including the transfer of the Division of Transplantation from HRSA to OASH will take longer to transition.” Statement of Organization, Functions, and Delegations of Authority ↩
-
See full governance map from Governance and Oversight in the Organ Donation Process,” The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
Revisions to the Outcome Measure Requirements for Organ Procurement Organizations; Final Rule, CMS, 2020. Table 5, pg 136. ↩
-
“It is clear that our historical approach to measuring OPO performance has resulted in a wide range of performances. This variability is unacceptable to patients and CMS. Thus, CMS intends to hold these entities to revised and higher standards. These revised metrics are necessary in light that over the past 14 years, the sharing of best practices, if it has occurred, has not resulted in consistent improvements throughout the industry.” Revisions to the Outcome Measure Requirements for Organ Procurement Organizations; Final Rule, CMS, 2020. ↩
-
“CMS Finalizes Policy that will Increase the Number of Available Lifesaving Organs by Holding Organ Procurement Organizations Accountable through Transparency and Competition” CMS, 2020. ↩
-
NASA’s FY 2020 Budget is $22.629 billion ↩
-
“Organ Donation Reform Money Flow Map,” The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
“Reforming Organ Donation in America,” Bridgespan, 2019. Updated projections, Bridgespan, 2020. ↩
-
“A Cost-Benefit Analysis of Government Compensation of Kidney Donors,” Am J Transplant, 2019. ↩
-
“Reforming Organ Donation in America,” Bridgespan, 2019. Updated projections, Bridgespan, 2020. ↩
-
Definition from “Organ Procurement and Transplantation Network (OPTN),” US Department of Health and Human Services. ↩
-
“Transplant monitor lax in oversight,” LA Times, 2006. ↩
-
[USC02] 42 USC 274: Organ procurement and transplantation network ↩
-
See several instances of this outlined in Appendix B ↩
-
“The OPTN is not authorized to monitor or enforce CMS Conditions for Coverage.” Attachment A UNOS Responses to February 10, 2020 Letter ↩
-
§121.10 (b1) of the Final Rule requires that the OPTN “design appropriate plans and procedures, including survey instruments, a peer review process, and data systems, for purposes of” (iii) Conducting ongoing and periodic reviews and evaluations of each member OPO and transplant hospital for compliance with these rules and OPTN policies.” Given the parallel language used in the Final Rule, it is unclear why the OPTN seems to have chosen to be aggressive on transplant hospital oversight but lax on OPO oversight. ↩
-
“UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin,” 2020. ↩
-
“Membership and Professional Standards Committee - OPTN” ↩
-
As UNOS explains, “[t]he MPSC’s work to define the process for OPO Performance review involved a work group consisting of all the MPSC members who worked at an OPO.” UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
Failing OPOs who are represented on the MPSC include LifeNet VA, Legacy of Hope, and Donor Alliance. Membership and Professional Standards Committee - OPTN ↩
-
“Transforming Organ Donation in America,” Table A, page 46. Bridgespan, 2020. ↩
-
Indeed, while the data HHS released in the Final Rule shows more than half of all OPOs to be underperforming, all 57 OPOs are in compliance per the OPTN, calling into question the rigor of these OPTN standards. ↩
-
See Appendix B, response to question 3a. ↩
-
December 18, 2019 VIA ELECTRONIC TRANSMISSION Ms. Joanne M. Chiedi Acting Inspector General Office of Inspector Genera ↩
-
NOTA’s structure allows UNOS to operate as a monopoly. In fact, more than twenty years ago, Forbes referred to UNOS as a “cartel” and “the federal monopoly that’s chilling the supply of transplantable organs and letting Americans who need them die needlessly.” From “The Organ King,” Forbes, 1999. As one transplant surgeon noted: “The OPTN contractor has devolved into a hostage-taking situation, where it has convinced the government that no one else can do what it does, but it doesn’t perform its functions particularly well.” ↩
-
“Technology Recommendations · The Costly Effects of an Outdated Organ Donation System,” 2020. ↩
-
According to one OPO leader, board members can be paid for services they offer outside of the board duties, in accordance with the bylaws. These services are decided by the OPO, and there’s nothing that prevents OPOs from adding this to their cost reporting. ↩
-
“Tier 3 OPOs will be considered as failing the outcome measures and will be de-certified.” https://www.cms.gov/files/document/112020-opo-final-rule-cms-3380-f.pdf ↩
-
OneLegacy Tax Return, ProPublica, page 7. ↩
-
“Review of OneLegacy’s Reported Fiscal Year 2006 Organ Acquisition Overhead Costs and Administrative and General Costs,” HHS, Office of Inspector General, 2010. ↩
-
“The 100 percent reimbursement for kidney costs creates incentives for cost-shifting, as OPOs have a financial interest in showing Medicare that their kidney-recovery costs exceed their reimbursements. Particularly for indirect costs (e.g., overhead, management), OPOs have the incentive to allocate as many costs as possible to kidney recovery rather than spreading them across multiple organ categories.” Transforming Organ Donation in America, Bridgespan, 2020. ↩
-
Whistleblowers have told us that some OPO CEOs, such as Tom Mone, often flaunt such political access afforded through the connections of their paid board members. HHS should evaluate the extent to which this practice occurs and whether such miscatergorization of expenses may contsitute Medicare Fraud. ↩
-
According to an analysis by the Associated Press of 2017 data, “Where you die can affect your chance of being an organ donor,” if OneLegacy recovered donors at the same rate as the OPO based in Philadelphia, it would have recovered 379 more donors in that year alone [note: this likely would have translated into roughly 1,000 more lives saved] ↩
-
Global Liver Institute Responds to AOPO’s Recent Misinformation Campaign. Donna Cryer, 2019. ↩
-
Improvements in organ donation: Riding the coattails of a national tragedy. Clinical Transplantation, 2019. ↩
-
Organ Procurement Money Flow Map. The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
“i. The OPO, Sharing Hope South Carolina (sending the wrong lungs, leading to the recipient’s death); ii. The OPO, Life Alliance Organ Recovery Agency (failing to identify an infection in a uterus it recovered for transplant, leading to a near-fatal outcome for the recipient); and iii. The OPO, Donor Network of Arizona (wrongfully recovering corneas from a donor who was not registered and whose family did not provide consent).” Senate Finance Committee Letter to HHS, 2020. ↩
-
“Transplant surgeon acquitted,” LA Times, 2008. ↩
-
Congressional Letter from Katie Porter and Karen Bass to HHS, 2020. ↩
-
“This litany of reports is an indictment against the industry and demonstrates the need for reform.” https://oversight.house.gov/news/press-releases/oversight-subcommittee-launches-investigation-into-poor-performance-waste-and ↩
-
“How Lifesaving Organs For Transplant Go Missing In Transit,” Kaiser Health News, 2020. ↩
-
“He died when he got the wrong lungs. It wasn’t the only organ error in SC that day,” Post and Courier, 2020. ↩
-
“Whistleblower threatened with being cremated alive after exposing mortuary kickback scheme,” CBS News, 2019. ↩
-
“Transforming Organ Donation in America,” The Bridgespan Group, 2020. ↩
-
William Chapman et al., “Medicare and Medicaid Programs; Organ Procurement Organizations Conditions for Coverage; Revisions to the Outcome Measure Requirements for Organ Procurement Organizations,” Public Comment to CMS, February 20, 2020. ↩
-
According to Congressional staffers, a common OPO strategy is to bring donor families into meetings “to try to make it uncomfortable for staffers to dig in on tough questions. I mean, it’s hard to really grill them about financial impropriety when they bring a donor mom to cry in front of me. Really, it’s just exploitative.” ↩
-
“11,000 Americans Will Die Waiting for Transplants This Year,” NY Times, 2019. ↩
-
Despite low performance, organ collection group gets new federal contract” Washington Post, 2019. ↩
-
https://obamawhitehouse.archives.gov/sites/default/files/omb/assets/oira_0938/0938_10292013b-1.pdf ↩
-
ARKANSAS REGIONAL ORGAN RECOVERY AGENCY v. Shalala, 104 F. Supp. 2d 1084 (E.D. Ark. 2000) ↩
-
Letter from former HHS Chief Technology Officer to HHS Secretary Alex Azar, 2019, citing quotations from OPO executives to The Bridgespan Group, “CMS is not engaged. They evaluate OPOs every 3-4 years, and the evaluation is a joke.” “The [OPO] system is in-bred and out of regulatory control.” “The OPO strategy is to confuse people with data – always have data available to tell your board or CMS that you’re doing good…‘As long as I’ve got something, I’m good.’” ↩
-
“How Lifesaving Organs For Transplant Go Missing In Transit,” Kaiser Health News, 2020. ↩
-
“Between 2014 and 2019, even only among the small subset of organs for which transportation was managed by the UNOS Organ Center, nearly 170 organs could not be transplanted and almost 370 endured “near misses,” with delays of two hours or more, after transportation problems.” Id. ↩
-
Code of Federal Regulations § 486.346 ↩
-
According to sources within the government ↩
-
UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
We define “shadow deaths” as people who die while on the waitlist because of missed opportunities for organs that were not recovered or transplanted, rather than directly resulting from a fatal patient safety lapse. ↩
-
https://www.renalandurologynews.com/home/news/nephrology/transplantation/with-donor-kidneys-every-hour-of-cold-ischemia-time-matters/ ↩
-
In another very similar case, this court document notes that “Upon receiving and examining the donated kidney, however, Hospital staff observed that it “was hard and cold to the touch, with patchy discoloration that appeared to resemble ‘freezer burn,’’ and determined that it was not suitable for transplant. The Hospital canceled Ms. Scarbrough’s transplant surgery.” Karen Scarbrough, the patient who was supposed to receive the frozen kidney, sued the OPO for negligence, asserting that the OPO staff “were negligent with respect to the collection, packaging, preservation, transportation, and/or handling of the donated kidney which resulted in irreversible damage to the kidney making it unsuitable for transplant.” Scarbrough v. Transplant Resource Center of Maryland, No. 815, September Term, 2018. Opinion by Fader, CJ MARYLAND REVISED UN ↩
-
_ “OPOs are funded on a cost-reimbursement basis, with Medicare and transplant centers covering 100 percent of costs for activities related to organ procurement. This arrangement appears to be unique in US healthcare.” From “Transforming Organ Donation in America,” Bridgespan, 2020. _ ↩
-
§ 486.326 All OPOs must have a sufficient number of qualified staff, including a director, a medical director, organ procurement coordinators, and hospital development staff to obtain all usable organs from potential donors, and to ensure that required services are provided to families of potential donors, hospitals, tissue banks, and individuals and facilities that use organs for research. Conditions for Coverage, CMS. ↩
-
Underperforming in New York: Nonprofit struggles to recover transplantable organs, Washington Post, 2019. ↩
-
UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
“Variables such as race and sex of OPO representative and time of day should be considered before approaching a family for organ donation. Avoiding translators during the approach process may improve donation rates.” From “Improving organ donation rates by modifying the family approach process,” The Journal of Trauma and Acute Care Surgery, 2014. ↩
-
“The impact of race on organ donation authorization discussed in the context of liver transplantation,” Transactions of the American Clinical and Climatological Association, 2012. ↩
-
“Communicating Effectively About Organ Donation: A Randomized Trial of a Behavioral Communication Intervention to Improve Discussions About Donation,” Transplantation Direct, 2015 ↩
-
“We heard of one example of an OPO that had a large Spanish-speaking population in the DSA, yet 9 out of 10 staff members could not speak Spanish.” OPO Best Practices · The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
“How a surgeon helped solve the problem of far too few black organ donors,” Center for Health Journalism, 2018. ↩
-
UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
Language in the OPTN Final Rule, which supersedes OPTN Bylaws, shows that peer-review protections are not nearly as broad as UNOS makes them out to be. Section 121.11, note that the HHS Secretary has the ability to view any documentation necessary, “All OPOs… shall maintain such records pertaining to… transplantation-related matters as the Secretary deems necessary to carry out her/his responsibilities under the Act.” OPTN Final Rule Language ↩
-
“The medical peer review privilege will not be extended to withhold any document from the Secretary of HHS, or the Secretary’s designee.” Pg 198, OPTN Bylaws effective Aug 17 2020 WORD [Updating Appendix D-7-B] ↩
-
https://www.casemine.com/judgement/us/59147b44add7b04934419d5c ↩
-
Nevada Donor Network and Life Alliance Organ Recovery Agency (Miami) are the two OPOs that have received “member not in good standing” designations, according to UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
“At the time of McFarland’s transplant, LAORA was a “member not in good standing” of the transplant network — a designation that indicated a “serious lapse in patient safety or quality of care,” according to UNOS. The reasons for that designation are secret, however. The designation, revealed only by a few paragraphs on UNOS’ website, triggered additional monitoring and other corrective measures, but did not stop LAORA from functioning.” “Why did the first US uterine transplant fail?” Washington Post, 2019. ↩
-
The same OPO, LAORA, was involved in alleged workplace violence. However, UNOS does not disclose why OPOs earn designations like “member not in good standing,” so it’s unclear if that was the cause. “Doctor, nurse: UM administrator assaulted us,” Miami Herald, 2015. ↩
-
UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
Example best practice from OSC: Guidance on Effective Practices in Broader Distribution, OPTN. ↩
-
Interface screenshots for this system can be found here: ASPEN 9.5 - CLIA AO Upload User Guide_v0.05 ↩
-
As outlined in the Bridgespan report, not having external checks on OPO finances has further implications. Not only does the current CMS reimbursement structure allow for conflicts of interest and abuses, it also structurally incentivizes misallocation of resources which ultimately affects organ procurement. The Bridgespan Group, Transforming Organ Donation in America, 2020. ↩
-
“Why did the first US uterine transplant fail?” Washington Post, 2019. ↩
-
“Doctor, nurse: UM administrator assaulted us,” Miami Herald, 2015. ↩
-
“University of Miami organ bank fails to meet regulatory requirements,” Miami Herald, 2015. ↩
-
“Alabama Organ Center kickbacks detailed,” Al.com, 2012. ↩
-
Analysis done by UPenn using state inpatient databases; cited in “Reforming Organ Donation in America,” Bridgespan, 2019. ↩
-
Review of OneLegacy’s Reported Fiscal Year 2006, Office of Inspector General OIG, 2010. ↩
-
“How organ and tissue donation companies worked their way into the county morgue,” LA Times, 2019. ↩
-
“Executives coached coroners on how to keep body parts harvesting records secret,” LA Times, 2019. ↩
-
“He died when he got the wrong lungs. It wasn’t the only organ error in SC that day.” Post and Courier, 2020. ↩
-
“Mesa family takes legal action after Donor Network of Arizona ‘wrongly harvests’ their son’s corneas,” ABC15.com, 2019. ↩
-
“Tissue donation turns into nightmare for three sisters,” Review Journal, 2016. ↩
-
Donatelife.net, Waiting list statistic. ↩
-
OPTN Board of Directors; OPOs represented include LifeChoice Donor Services (tier 2), Iowa Donor Network (tier 3), and Louisiana Organ Procurement Agency (tier 2) according to data from 2018; tier 3 will be decertified and tier 2 up for competition and could lose their contracts ↩
-
Revisions to the Outcome Measure Requirements for Organ Procurement Organization, HHS, 2020. ↩
-
Failing OPOs represented on the MPSC include: Colorado’s Donor Alliance (also serves part of Wyoming), Alabama’s Legacy of Hope, and Virginia’s LifeNet Health. Membership and Professional Standards Committee - OPTN ↩
-
As UNOS explains, “[t]he MPSC’s work to define the process for OPO Performance review involved a work group consisting of all the MPSC members who worked at an OPO.” UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
Senate Finance Committee Letter to the Office of Inspector General, 2019. ↩
-
Government officials have noted that they now write “OPTN/UNOS” in all formal communications, rather than addressing anything to just OPTN or UNOS individually, “so that there is no wiggle room for UNOS to evade a question by saying, ‘well we did such-and-such a thing in our UNOS capacity, and your question pertains OPTN.’” ↩
-
The former UNOS president, James Burdick, became HRSA’s Department of Transplantation (DoT) director. And DoT Director Melissa Greenwald left HRSA to go to an OPO. ↩
-
Callahan v. U.S. Department of Health and Human Services, Case No. 1:19-cv-01783-AT (D. Ga.). In any earlier Court Order in the same case, the Judge wrote that she was “troubled by [UNOS’s] lack of candor. ↩
-
“How organ and tissue donation companies worked their way into the county morgue,” LA Times, 2019. ↩
-
In the same year, the LifeLink Foundation CEO, Jean Davis, nearly doubled her taxpayer-funded compensation from $695,031 in 2018 to $1,235,941 in 2019, and the Board Chair, Dennis Heinrichs, increased his from $748,460 to $919,066, underscoring the incentive for LifeLink to lobby against decertifications for failing OPOs. ↩
-
House Committee on Oversight and Reform, Investigation into LifeLink Foundation, December 2020 ↩
-
Heartless: Organ Donation Contractors Lobby Against a Popular Health Care Initiative While Pocketing Pandemic Relief Loans. Project on Government Oversight, 2020. ↩
-
https://oversight.house.gov/news/press-releases/oversight-subcommittee-launches-investigation-into-poor-performance-waste-and ↩
-
Committee to address geographic aspects of organ distribution, OPTN ↩
-
In a court filing on December 12, 2019, UNOS claimed that Glazier was even acting as UNOS’ counsel – subject to attorney-client privilege – compromising UNOS’ motivation and ability to exercise its oversight responsibilities over her OPO. ↩
-
New England Donor Services Leadership Team via neds.org/leadership-team ↩
-
New England Donor Services Leadership Team via neds.org/leadership-team ↩
-
A Fairer and More Equitable, Cost-Effective, and Transparent System of Donor Organ Procurement, Allocation, and Distribution, The National Academies.org ↩
-
“Despite low performance, organ collection group gets new federal contract”, Washington Post, 2019. ↩
-
While there have been efforts such as the Organ Donation Breakthrough Collaborative, there is not a sustained and systematic way that best practices are collected, verified, and disseminated. ↩
-
UNOS Response to 2.10.20 Letter from Sens Grassley Wyden Young and Cardin, 2020. ↩
-
The Costly Effects of an Outdated Organ Donation System: Money Flow Map, 2020. ↩
-
The Bridgespan Group, Transforming Organ Donation in America, 2020. ↩
-
“Taxpayers help pay for organ donor groups’ parties, Rose Parade expenses,” Trib Live, 2013. ↩
-
“The Costly Effects of an Outdated Organ Donation System,” The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
Revisions to the Outcome Measure Requirements for Organ Procurement Organization, CMS, 2019. ↩
-
42 CFR Part 486 - CMS OPO Final Rule, CMS, 2020. ↩
-
While lobbying expenses are technically not reimbursable for OPOs, a dishonest OPO could spend money on lobbying, and not be directly reimbursed for it, but raise its SAC fees to recoup the money and net out even. ↩
-
Heartless: Organ Donation Contractors Lobby Against a Popular Health Care Initiative, Project on Government Oversight, 2020 ↩
-
Heartless: Organ Donation Contractors Lobby Against a Popular Health Care Initiative, Project on Government Oversight, 2020 ↩
-
Heartless: Organ Donation Contractors Lobby Against a Popular Health Care Initiative, Project on Government Oversight, 2020 ↩
-
There is congressional oversight interest in this matter. The House Committee on Oversight and Reform (Subcommittee on Economic and Consumer Policy) recently issued a letter to AOPO requesting documentation on its lobbying efforts related to CMS’s proposed OPO rule changes. https://oversight.house.gov/news/press-releases/oversight-subcommittee-launches-investigation-into-poor-performance-waste-and ↩
-
OneLegacy Complaint - See more cases in Appendix A ↩
-
“An informal survey conducted of four representative organ procurement organizations shows the percentage of organ donors that ultimately donate tissue: OneLegacy (Los Angeles): 55%; Lifesource (Minn, SD, ND): 40%; LifeNet Health (Virginia): 45%; Transplant Resource Center of Maryland: 25%” https://www.notifylibrary.org/sites/default/files/BOOK%20NOTIFY.pdf,” World Health Organization, 2011. ↩
-
“How organ and tissue donation companies worked their way into the county morgue,” LA Times, 2019. ↩
-
Congressional oversight has a strong interest in this issue. The House Committee on Oversight and Reform (Subcommittee on Economic and Consumer Policy) recently issued letters to OPOs requesting documentation on conflicts of interests with tissue recovery and misuse of funds and the Ways and Means Oversight Committee recently issued a letter to HHS inquiring about oversight of OPO financial incentives, including the question, “What specific processes does HHS have in place to ensure that conflicts are addressed when they arise, and that patients are not unduly pressured to donate tissue?” https://oversight.house.gov/news/press-releases/oversight-subcommittee-launches-investigation-into-poor-performance-waste-and ↩
-
“She Beat Cancer. Now She’s in Another Fight for Her Life,” New York Times, 2019. ↩
-
“Summary of Findings · The Costly Effects of an Outdated Organ Donation System” The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
“Technology Recommendations · The Costly Effects of an Outdated Organ Donation System,” The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
“Technology Recommendations · The Costly Effects of an Outdated Organ Donation System,” The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
“Summary of Findings · The Costly Effects of an Outdated Organ Donation System,” The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
Addressing the Organ Donor Crisis, Dayoneproject.org. ↩
-
Several procurement recommendations are outlined in this report: Strategy for Buying OPTN Tech · The Costly Effects of an Outdated Organ Donation System, The Costly Effects of an Outdated Organ Donation System, 2020. ↩
-
We recommend defining conflict of interest here as any financial, professional, or personal ties to any of the parties involved in the conflict or their partners (such as foundations, other business entities with shared ownership, etc), including if the MPSC recommended actions to the OPO that financially benefited the MPSC member or their partners in anyway. ↩
-
Currently, CMS can decertify an OPO at any time for “Urgent Need”, which it defines as “occur[ing] when an OPO’s non-compliance with one or more conditions for coverage has caused, or is likely to cause, serious injury, harm, impairment, or death to a potential or actual donor or an organ recipient.” However, it is unclear what “serious injury, harm, impairment or death” an OPO can cause to an “actual donor”, who, by definition, would already be deceased. While applying this standard to an “organ recipient” is reasonable, CMS should consider expanding Urgent Need to apply to waiting list patients as well, reflecting the reality that most deaths caused by OPOs are “shadow deaths.” ↩
-
“The current OPO payment model does not give OPOs an incentive to reallocate resources in order to increase the number of organs available for transplant and reimburses OPOs for costs which may in fact not help produce the desired outcomes.” From “Transforming the Organ Donation System in America,” Bridgespan, 2020. ↩
-
The Government Accountability Office (GAO) has said that UNOS must do this as part of their renewed contract, but based on publicly available information, it does not appear that UNOS has separated their boards yet. https://www.gao.gov/assets/700/693614.pdf ↩
-
Table 4, Transplant Center Regulations—A Mixed Blessing? An ASTS Council Viewpoint ↩
-
OPTN/SRTR Annual Data Report 2018: American Journal of Transplantation: Vol 20, No s1 ↩
-
In July 2020, the Congressional Appropriations Committee, noting organ discard rates, wrote: The Committee supports HHS’ Request for Information for the technology system over which these organ offers are facilitated and encourages HHS to promote competition for this contract” ↩
-
https://www.finance.senate.gov/imo/media/doc/CEG.Young%20to%20HHSOIG%20(OPO%20Oversight)%20Dec.18.2019.pdf ↩
-
Thus far, despite multiple Congressional requests, the OPO has not provided any relevant information to prove or disprove these allegations, though whistleblowers have told us “the abuse was rampant.” Within the current financial structure, OPOs can pass through expenses to payors to cover their operations as part of the organ acquisition fees. ↩
-
In December 2018, Senator Young told the Washington Post “We can’t continue to allow [thousands of] Americans to die each year waiting for lifesaving organs that we know are available if only this system were being managed by competent individuals operating in the light of day,” (available at https://www.washingtonpost.com/graphics/2018/national/organ-transplant-shortages/) ↩